Page Outline
- Introduction: What makes a Lipid a Lipid? They’re hydrophobic!
- Fats and Oils
- Fats and Oils Quiz
- Phospholipids
- Steroids
- Waxes
- Lipids Quiz
1. Lipids are hydrophobic
Lipids
make up a diverse group of biomolecules that include
- fats and oils (used for energy storage and insulation by a variety of organisms);
- phospholipids (a key component of cell membranes);
- steroids (a molecule found in many hormones); and
- waxes (used for waterproofing, particularly on the surfaces of leaves)
What makes all of these types of molecules lipids is that they are either completely hydrophobic (which means “water-fearing”), or have large hydrophobic regions.
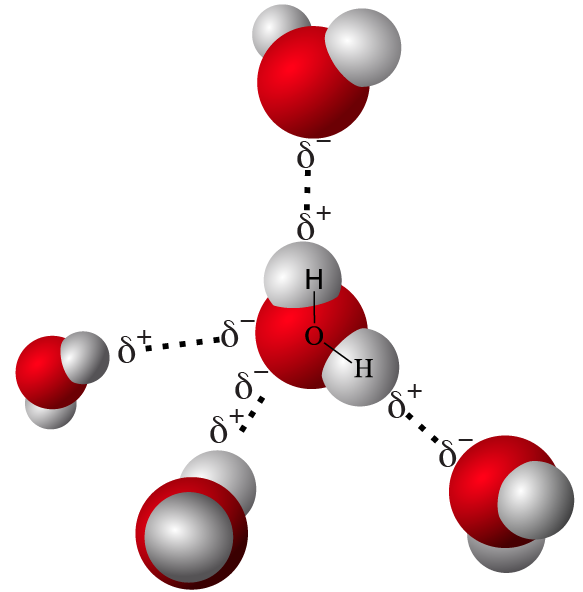
Being hydrophobic has to do with how a molecule interacts with water. Water (as you can review here) is a polar molecule. Polar molecules have unequal electron sharing, which results in the molecule having partially positively charged (designated as δ+ in the diagram to your left) and partially negatively charged (δ–) regions. These charged regions interact with one another in one of two ways: similarly charged regions repel one another, and oppositely charged regions attract one another. The attraction between positively and negatively charged regions of water molecules is called a hydrogen bond (indicated by the dotted lines to the left).
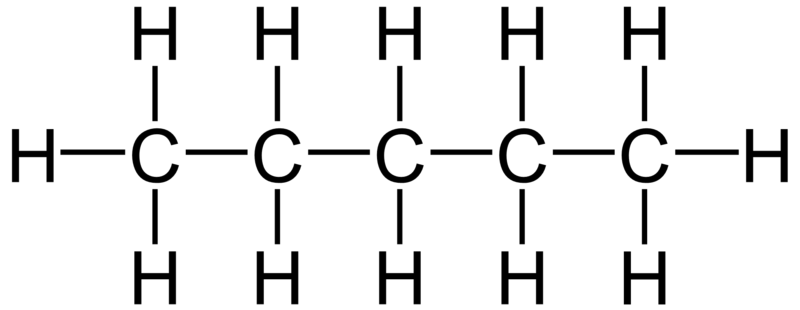
Hydrophobic molecules don’t dissolve in water. That’s because they’re non-polar. Take a look, for example, at pentane, the molecule to the right. Pentane is not a lipid, but we’ll see that the kind of molecular structure that it has a chain of carbon atoms surrounded by hydrogen atoms shows up in several types of lipids. This chain is called a hydrocarbon chain, and because electrons are shared evenly between the carbon and hydrogen atoms, there’s no polarity. This has two consequences. First, there’s nothing for water molecules to grab onto, so water can’t form hydrogen bonds with pentane. As a result, water can’t dissolve pentane (or any other non-polar molecule). Second, adjacent hydrophobic molecules will form very weak bonds with one another that cause them to cluster together. These bonds are caused by London dispersion forces: a very weak type of intermolecular bond, much weaker than the hydrogen bonds we’ve discussed above.
So, when you see oil droplets in water like this:

You should imagine this:
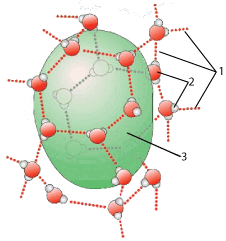
The green blob (at “3”) is a droplet of hydrophobic molecules clustering together because of their own very weak attraction to one another. At the same time, these hydrophobic molecules are prevented from dissolving in the surrounding solution of water molecules (two of which are indicated by “2”) because they don’t form hydrogen bonds (at “1”) with the water molecules in the solution.
Now that we understand the hydrophobic nature of lipids, let’s look at the four different types.
2. Fats and Oils
2a. The functions of fats and oils
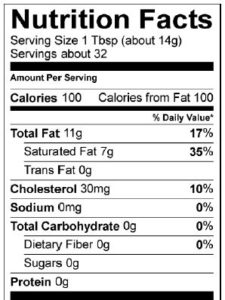
Fats and oils are the lipids that we eat. They’re the most energy-rich biomolecules. Take a moment and examine these photos and food labels for olive oil and butter.
 |
 |
|
|
|
Fats and oils both have about 9 food calories/gram. To put that in context, one tablespoon of oil or butter has 120 food calories. By contrast, carbohydrates have4 calories/gram. As we’ll see in the next tutorial, the same is true of proteins.

The calorie-dense nature of oils and fats explains one of their primary functions in living things: energy storage. In general, oils are synthesized by plants for energy storage, while fats are synthesized by animals (but there are plenty of exceptions). The oils in seeds (like peanut oil) store energy for the plant embryo. Animals that hibernate store the energy from the food they’ve eaten as fat, typically underneath the skin, but also in bone marrow or around internal organs.
This use of fat for energy storage is true of humans and other primates, too. If we take in more food energy than we need, we’ll first store it as the polysaccharide glycogen, which gets loaded into muscle and liver tissue (you can read more about this in the previous tutorial). However, our capacity to store glycogen is limited to only a small percentage of our body weight. Any additional food energy gets converted into fat (which can be stored in unlimited quantities).
Another function of fat is insulation. Think of marine mammals like whales, dolphins, seals, sea lions; or penguins. All of these animals have thick layers of blubber, a special form of insulating fat. Blubber also serves as a source of stored energy and makes marine animals more buoyant. If you’re interested, you can read more about blubber here.
A special tissue called brown fat is used for thermoregulation (controlling body temperature). The brown color comes from the fact that this tissue is loaded with mitochondria. It’s found in particularly high abundance in babies and hibernating mammals. You can read more about it here.
2b. Fats and oils are both triglycerides, made from glycerol and fatty acids
Oils are liquid at room temperature. Fats, found in lard (animal fat), shortening, margarine, butter, cheese, and cream, are solid at room temperature. The “room temperature” part is important. It’s easy enough to get oils to solidify when you put them in a refrigerator, and all you have to do is to put butter on a frying pan to get it to liquify.
Unlike polysaccharides, lipids aren’t composed of hundreds of repeating monomers. But they do have building blocks. The most important of these are fatty acids.

Palmitic acid (C16H32O2, shown above) is a fatty acid commonly found in animals, plants, and bacteria. Like all fatty acids, it has two key parts. At “1” there’s a long hydrocarbon chain. That’s the part with all of the chemical energy, with the energy stored in the bonds between the carbon atoms, and between the carbon and hydrogen atoms. At “2” is a carboxylic acid functional group (click here to review functional groups). The carboxylic acid can donate a proton to the solution that it’s dissolved in, making that solution more acidic (which is why this is a fatty acid).
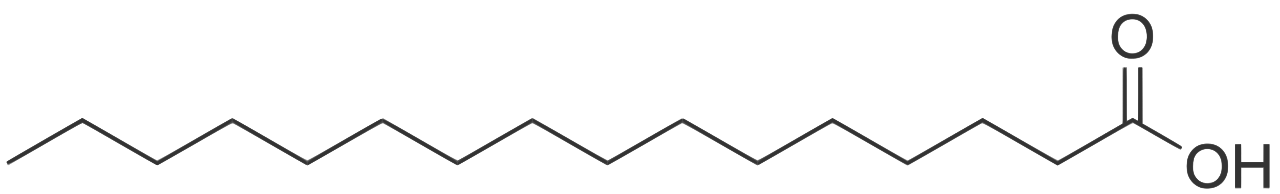
Here’s a skeletal structural formula for palmitic acid. (Remember that at every angle vertex there’s a carbon atom, and assume that each of the carbons in the hydrocarbon chain is also attached to as many hydrogen atoms as possible)
Fatty acids can be saturated or unsaturated.

Saturated means “full of moisture: made thoroughly wet” (Merriam-Webster). When a sponge is saturated with water, it’s holding as much water as it possibly can. In a saturated fatty acid, like the palmitic acid shown above, each carbon in the hydrocarbon chain is saturated with hydrogen and is bonded to as many hydrogen atoms as it possibly can hold on to. Another way of saying this is that there are no double bonds in the hydrocarbon chain (part “1”).
Here’s an unsaturated fatty acid. This is linoleic acid, commonly found in nuts and seeds.

Note the double bonds (indicated by arrows). The double bonds make the fatty acid unsaturated.
Here’s the skeletal structural formula for linoleic acid. You know that it’s unsaturated because of the double bond in the hydrocarbon chain after the 9th and 12th carbons.
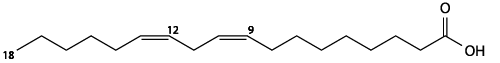
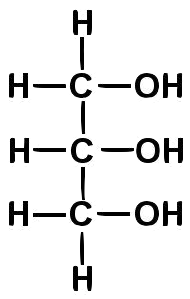
Oils and fats (like the olive oil and butter above) are composed of molecules called triglycerides (also known as triacylglycerides). Triglycerides consist of three fatty acids bonded to a three-carbon alcohol called glycerol (shown at right).
Fatty acids get linked to glycerol through dehydration synthesis reactions, as shown below.
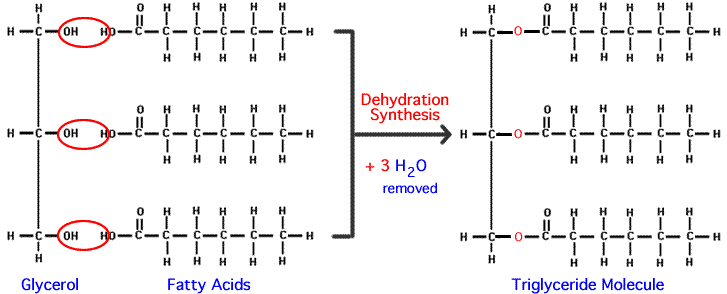
So, what makes fats and oils different? Why are fats solid at room temperature, and oils liquid? It’s all about the fatty acids within the triglyceride.
In fats, the three fatty acids are saturated.
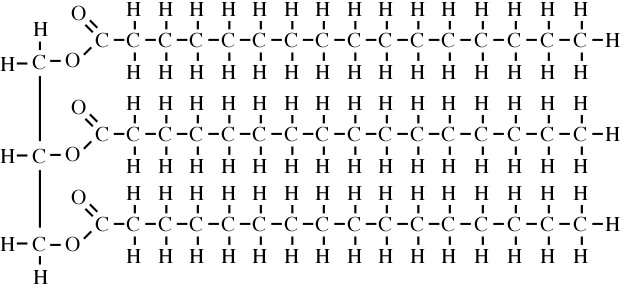
 Notice that in saturated fatty acids, the hydrocarbon chains are straight. This allows the fatty acids to form very weak bonds with one another (through London dispersion forces), as well as with adjacent triglyceride molecules. These bonds keep the resulting substances (like shortening, butter, margarine, or fat) solid at room temperature. The bonds enable them to hold their shape.
Notice that in saturated fatty acids, the hydrocarbon chains are straight. This allows the fatty acids to form very weak bonds with one another (through London dispersion forces), as well as with adjacent triglyceride molecules. These bonds keep the resulting substances (like shortening, butter, margarine, or fat) solid at room temperature. The bonds enable them to hold their shape.
In oils, at least one of the fatty acids within the triglyceride molecule is unsaturated, as shown below.
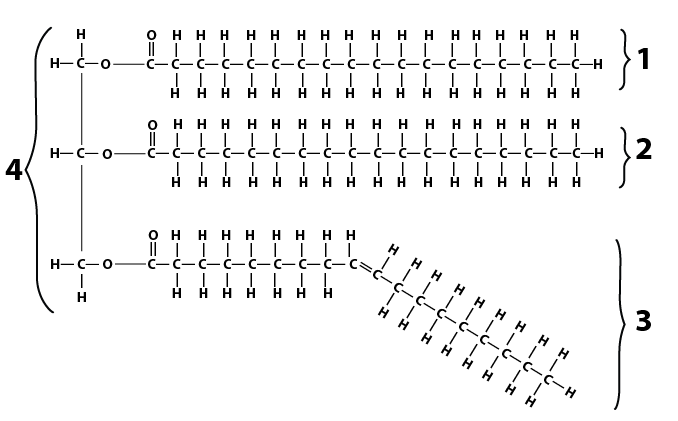
 The double bond in the third fatty acid causes the hydrocarbon chain to bend. Because of this bend, the fatty acids can’t bond with one another, or with other oil molecules. Because there’s no bonding, the molecules can’t hold their shape, making them liquid.
The double bond in the third fatty acid causes the hydrocarbon chain to bend. Because of this bend, the fatty acids can’t bond with one another, or with other oil molecules. Because there’s no bonding, the molecules can’t hold their shape, making them liquid.
Note that in all the diagrams above, I’ve shown the triglycerides with all of their fatty acids on the same side, lined up like people with their arms held at their sides. But in molecules with single bonds, the atoms can rotate around the single bond. That means that an unsaturated triglyceride could just as easily look like this:
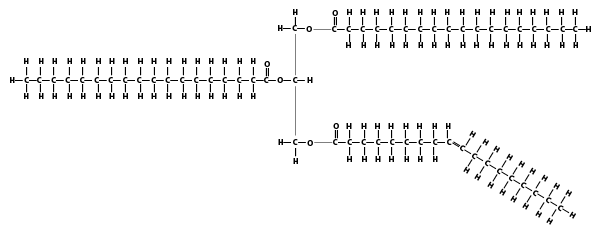
2c. Saturated Fats, Trans fats, and Cardiovascular Health
According to the American Heart Association, it’s a good idea to limit saturated fats to only a small proportion of the calories you take in. That’s because saturated fat intake seems to be connected with heart disease. The Heart Association’s recommendation is to limit saturated fats to about 13 grams/day. (source: American Heart Association) Here’s the saturated fat content of a few fatty foods.
| Food (and quantity) | Total Fat | Saturated Fat | Saturated fat density (saturated fat grams/gram of food) |
| Dry Salami (1 oz/28 grams) | 11 grams | 4 grams | 0.25 |
| Ground Beef (4 oz/113 grams) | 17 grams | 7 grams | 0.06 |
| Cheddar Cheese Stick (3/4 oz/21 grams) | 7 grams | 4.5 grams | 0.21 |
| Breakfast sausage (4 oz/113 grams) | 20 grams | 8 grams | 0.07 |
In a typical North American diet, saturated fat can build up pretty fast. If you had sausage for breakfast and a cheeseburger for lunch, you’d be over the American Heart Association recommendations. Note also that the type of meat you choose is important. The ground beef that I listed above is 85% lean meat, and 15% fat. There are both fatter and leaner types of meat.
There’s also one kind of unsaturated fat to watch out for. These are called trans fats. Here’s how they work.
I showed above how pieces of molecules can freely rotate around single bonds. That’s not true of double bonds. For example, look at the fatty acid below:

This is linoleic acid. As mentioned above, it’s a common fatty acid in nuts and seeds. It has two double bonds. In both of these bonds, the hydrogen atoms are on the same side of the molecule. In bond “a” both of the hydrogens are locked in place below the hydrocarbon chain, and in bond “b” both of the hydrogens are locked in place above the hydrocarbon chain (keep in mind, of course, that the entire molecule could rotate, but I’m hoping you get the point: the hydrogens in each double bond are on the same side of the chain).
When both hydrogens are on the same side of the chain, it’s called a cis configuration. So, this fatty acid is called cis-linoleic acid.

Contrast this with Elaidic acid.
There’s only one double bond. Note how one of the hydrogens is above the hydrocarbon chain, and the other is below the hydrocarbon chain. This is the trans configuration. To remember this, just remember that the prefix trans (as in transport, transgender, or trans-Siberian) means “across.”(dictionary.com) Trans fatty acids occur rarely in nature (they’re found in small amounts in milk and meat products). But they’re generated in large amounts in a process called hydrogenation, an industrial process where hydrogen atoms are forced into unsaturated vegetable oils, making them more saturated. This increases their shelf life and makes them easier to use in processed foods.
Trans fats, like the Elaidic acid shown above, have been implicated in contributing to heart disease. Click here to read what the American Heart Association has to say about trans fats.
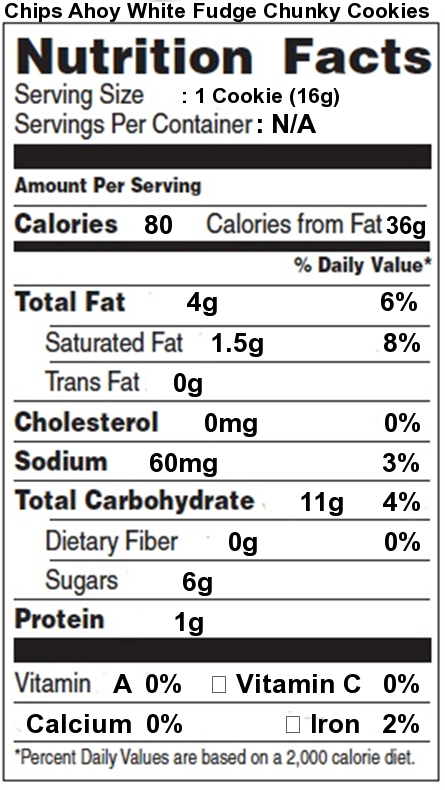 Food companies have become sensitive to consumer concerns over the connection between saturated fats, trans fats, and health. Take a minute to look at the nutrition facts label for Chips Ahoy White Fudge Chunky cookies. As with almost any food with a nutrition facts label, you can see total fat, saturated fat, and trans fat.
Food companies have become sensitive to consumer concerns over the connection between saturated fats, trans fats, and health. Take a minute to look at the nutrition facts label for Chips Ahoy White Fudge Chunky cookies. As with almost any food with a nutrition facts label, you can see total fat, saturated fat, and trans fat.
3. Fats and Oils: Checking Understanding
[qwiz use_dataset=”SMV_biochem5_fats_and_oils” dataset_intro=”false” qrecord_id=”sciencemusicvideosMeister1961-Fats and OIls Quiz (2.0)”]
[h]Fats and Oils: Checking Understanding
[i]
[x][restart][/qwiz]
4. Phospholipids
Phospholipids are the key components of cell membranes.
Phospholipids have three key features.
- Like triglycerides, phospholipids are built around a molecule of glycerol (shown in pink at number 2 in the diagram).
- Attached to the glycerol on one side are two fatty acids (shown in yellow at number 3). The two fatty acids make up the molecule’s tail.
- On the other side of the molecule (number 1 in the diagram) is a phosphate-bearing head.
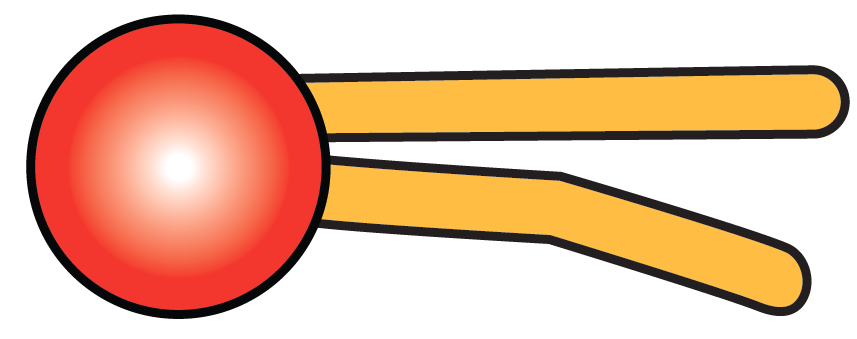
Typically, phospholipids are represented by diagrams like the one shown on the left. The fatty acid tails are on the right, in orange. The phosphate-bearing head is on the left, in red.
In terms of how phospholipids interact with water, they have a split personality.
- The fatty acid tails are non-polar, making them hydrophobic (water-fearing). In other words, they act like a fat or an oil molecule, avoiding water.
- The head is polar. This makes the head hydrophilic (water-loving). In other words, the head interacts with water by forming hydrogen bonds with whatever water molecules it bumps into.
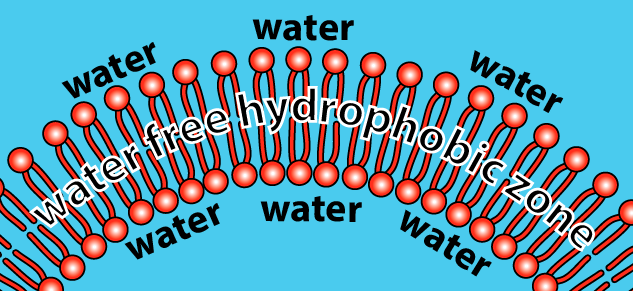
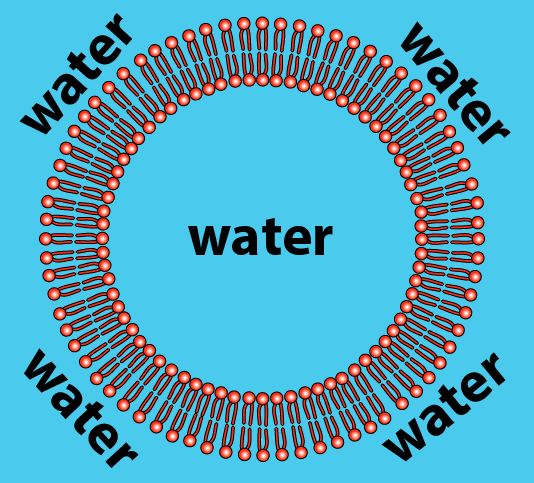 As a result, phospholipids, when mixed with water, spontaneously organize themselves into a few orientations. One is called a bilayer. The bilayer is a membrane that defines a sphere (which you can think of as an empty cell). If we sliced open the sphere, here’s what we’d see:
As a result, phospholipids, when mixed with water, spontaneously organize themselves into a few orientations. One is called a bilayer. The bilayer is a membrane that defines a sphere (which you can think of as an empty cell). If we sliced open the sphere, here’s what we’d see:
How does this work? Let’s just look at a slice of the sphere.
- The hydrophilic heads of the phospholipids orient themselves so that the heads are in contact with water.
- The hydrophobic tails cluster together. This is the same clustering that happens in a droplet of oil mixed in water. All of the tails create a hydrophobic, water-free zone. This zone is stabilized by weak bonds (hydrophobic interactions or Van der Waals forces) that occur between the tails.
We’ll see in Unit 2 that phospholipids are the basis of cell membrane structure in two of the three domains of life (Eukaryotes and Bacteria). The organisms in life’s third domain, the Archaea, use a different lipid molecule to build their cell membranes.
[qdeck random = “true” style=”width: 528px; border: 2px solid black; ” qrecord_id=”sciencemusicvideosMeister1961-Phospholipids Flashcards (2.0)”]
[h] Flashcards: Phospholipids and the phospholipid bilayer
[i]
[q]Using the terms polar, nonpolar, hydrophobic, hydrophilic (and other terms as needed), describe the structure of a phospholipid.
[a]Phospholipids have a hydrophilic, polar head and a nonpolar, hydrophobic tail. Two fatty acid chains make up the phospholipid’s “tail.” The head contains a negatively charged phosphate group, as well as other nonpolar components. Glycerol, the same 3-carbon alcohol found in triglycerides, connects the tail and head.
[q]When phospholipids are mixed with water, what happens?
[a]When mixed with water, phospholipids organize themselves into several configurations, one of which is a sphere that’s bounded by a phospholipid bilayer. The hydrophilic heads face out, interacting with water that’s outside or inside the sphere through hydrogen bonds. Within the bilayer, the hydrophobic phospholipid tails form a water-free zone that’s stabilized by hydrophobic interactions between the tails.
[q]In the diagram below, name regions 1, 2, and 3.
[a]In the diagram below, 1 is the hydrophilic head, 2 is glycerol, and 3 is the hydrophobic tail region.
[x]
If you want more practice, please press the restart button below. Otherwise, follow the links below.
[restart]
[/qdeck]
5. Steroids
The molecule below is testosterone, an example of a type of lipid called a steroid.
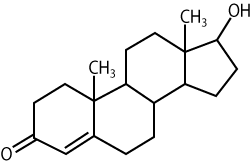
Steroids are all built around four or five fused carbon rings. What makes one steroid different from another are variations in this carbon skeleton (for example, the position of double bonds) along with the attachment of various chemical groups. See, for example, if you can find the difference between testosterone and the steroid molecule below.
[qwiz style=”width: 600px !important;” qrecord_id=”sciencemusicvideosMeister1961-Steroid Structural Formula”]
[h]Analyzing a steroid structural formula
[q]
[c]IENsaWNrIHdoZW4geW91JiM4MjE3O3JlIH JlYWR5IHRvIHNlZSB0aGUgYW5zd2VyLg==[Qq]
[f]
Cg==| [Qq]
|
|
The molecule on the left is estrogen. Compared to testosterone, estrogen has 1) two more double bonds in the carbon ring on the bottom left, 2) no methyl group(CH3) sticking up between the two bottom carbon rings, and 3) a hydroxyl functional group (OH) instead of a carbonyl group (=O) sticking out from the lower left. Those three differences, though, are enough to account for many of the biological differences between females and males.
[/qwiz]
As you can see from the example of testosterone and estrogen, one function of steroids is to act as hormones. Hormones are molecules that are produced in a gland in one part of the body, get released into the bloodstream, and, in the case of steroid hormones, enter into cells and cause changes in cellular activity (there are other types of hormones that don’t enter into cells, but which bind at the membrane).
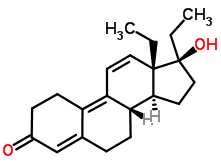
If you’re a sports fan, you’re probably aware of athletes who have been accused of and/or found guilty of using steroids to increase their athletic performance. These drugs are called anabolic steroids. You can think of them as artificial forms of testosterone, and they have the same effect on the body as testosterone has: increasing muscle mass, which increases strength and performance. One such performance-enhancing anabolic steroid is tetrahydrogestrinone, which was used by baseball player Barry Bonds (knowingly or not: you can read about the controversy here).
A steroid with a different role is cholesterol.
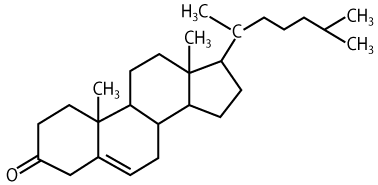
Cholesterol is found in cell membranes, where it keeps membrane molecules like phospholipids and proteins fluid (which is important for allowing diffusion of substances across membranes), Cholesterol also maintains membrane strength, which is important in animal cells, which lack cell walls. Cholesterol, which is synthesized primarily by the liver, is also a starting point for the biosynthesis of other steroid molecules, including the steroid hormones discussed above.
Cholesterol, along with the saturated fats and trans-fats discussed above, can negatively impact cardiovascular health. Click the link that follows to read what the American Heart Association has to say about cholesterol and heart disease.
6. Waxes
Waxes are another type of lipid.
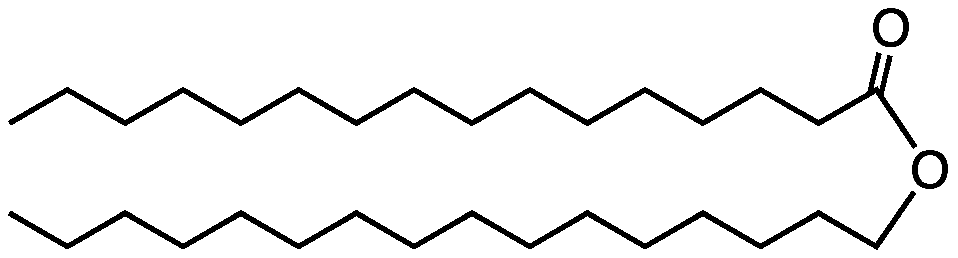
In plants, waxes are found on the upper surfaces of leaves, where they prevent water loss. The diagram of a leaf cross-section below shows this waxy layer (also called a “cuticle”) at number “1,” on both the upper and lower surface of the leaf.
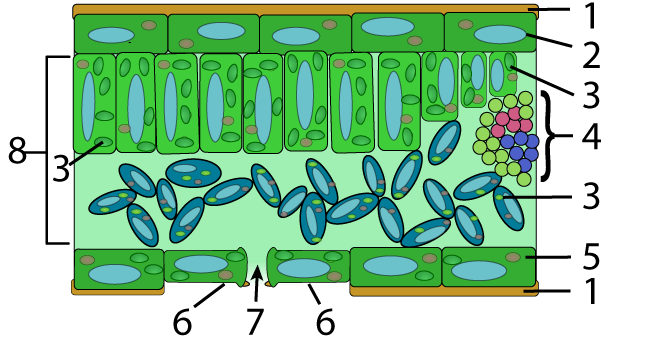
In the animal world, bees use wax to construct their honeycombs. Worker bees synthesize this wax in wax-producing glands in their abdomens that convert the sugar in honey into wax.

As with fats, bonds between the hydrocarbon chains within wax molecules and between wax molecules make waxes solid. And while the melting point of waxes is higher than that of fats like lard or margarine, it’s not that high (as anyone who has ever lit a wax candle knows).
7. Lipids: Checking Understanding
[qwiz qrecord_id=”sciencemusicvideosMeister1961-Lipids Quiz”] [h]
Lipids
[i]
[q labels = “top”]
[l]waterproofing
[fx] No, that’s not correct. Please try again.
[f*] Correct!
[l]membranes
[fx] No, that’s not correct. Please try again.
[f*] Correct!
[l]fats and oils
[fx] No, that’s not correct. Please try again.
[f*] Excellent!
[l]insulation
[fx] No. Please try again.
[f*] Good!
[l]steroids
[fx] No, that’s not correct. Please try again.
[f*] Great!
[q json=”true” multiple_choice=”true” xx=”3″ dataset_id=”SMV_biochem6_lipids_cumulative|8ce25c964a33b” question_number=”1″] The molecule shown below is a
[c]IGZhdHR5IGFjaWQ=[Qq]
[f]IE5vLiBGYXR0eSBhY2lkcyBoYXZlIGEgbG9uZyBoeWRyb2NhcmJvbiB0YWlsLCBhdHRhY2hlZCB0byBhIGNhcmJveHlsIGdyb3VwLiBOb3RlIHRoZSBmb3VyIGZ1c2VkIGNhcmJvbiByaW5ncy4=
Cg==[Qq]
[c]IHRyaWdseWNlcmlkZQ==[Qq]
[f]IE5vLiBUcmlnbHljZXJpZGVzIGhhdmUgdGhyZWUgZmF0dHkgYWNpZHMgYXR0YWNoZWQgdG8gb25lIGdseWNlcm9sLg==[Qq]
[c]IHdheA==[Qq]
[f]IE5vLiBXYXhlcyB1c3VhbGx5IGNvbnNpc3Qgb2YgdHdvIGxvbmcgaHlkcm9jYXJib24gY2hhaW5z
Cg==[Qq]
[c]IHN0ZX JvaWQ=[Qq]
[f]IEV4Y2VsbGVudC4gVGhpcyBtb2xlY3VsZSBpcyBhIHN0ZXJvaWQgKHNwZWNpZmljYWxseSwgaXQmIzgyMTc7cyB0aGUgaG9ybW9uZSBlc3Ryb2dlbik=[Qq]
[c]IHBob3NwaG9saXBpZA==[Qq]
[f]IE5vLiBQaG9zcGhvbGlwaWRzIGNvbnNpc3Qgb2YgdHdvIG5vbi1wb2xhciBmYXR0eSBhY2lkIGNoYWlucywgYXR0YWNoZWQgdG8gZ2x5Y2Vyb2wsIHdoaWNoIGlzIGF0dGFjaGVkIHRvIGEgcG9sYXIgJiM4MjIwO2hlYWQmIzgyMjE7IGNvbnNpc3Rpbmcgb2YgYSBwaG9zcGhhdGUgZ3JvdXA=
Cg==[Qq]
[q json=”true” multiple_choice=”true” xx=”2″ dataset_id=”SMV_biochem6_lipids_cumulative|8cddd9bf4073b” question_number=”2″] The molecule shown below is a
[c]IGZhdHR5IGFjaWQ=[Qq]
[f]IE5vLiBGYXR0eSBhY2lkcyBoYXZlIGEgbG9uZyBoeWRyb2NhcmJvbiB0YWlsLCBhdHRhY2hlZCB0byBhIGNhcmJveHlsIGdyb3VwIChhcyBzaG93biBiZWxvdykuIFRoZSBtb2xlY3VsZSBhYm92ZcKgaW5jbHVkZXM=IGZhdHR5IGFjaWRzIChidXQgaXQmIzgyMTc7cyBub3QganVzdCBhIGZhdHR5IGFjaWQp
Cg==[Qq]
[c]IHRyaWdseWNlcmlkZQ==[Qq]
[f]IE5vLiBUcmlnbHljZXJpZGVzIGhhdmUgdGhyZWUgZmF0dHkgYWNpZHMgYXR0YWNoZWQgdG8gb25lIGdseWNlcm9sLg==[Qq]
[c]IHdh eA==[Qq]
[f]IEV4Y2VsbGVudCEgV2F4ZXMgY29uc2lzdCBvZiB0d28gbG9uZyBoeWRyb2NhcmJvbiBjaGFpbnMu
Cg==[Qq]
[c]IHN0ZXJvaWQ=[Qq]
[f]IE5vLiBTdGVyb2lkcyBjb25zaXN0IG9mIGZvdXIgZnVzZWQgY2FyYm9uIHJpbmdzLCBhcyBzaG93biBiZWxvdy7CoA==[Qq]
[c]IHBob3NwaG9saXBpZA==[Qq]
[f]IE5vLiBQaG9zcGhvbGlwaWRzIGNvbnNpc3Qgb2YgdHdvIG5vbi1wb2xhciBmYXR0eSBhY2lkIGNoYWlucywgYXR0YWNoZWQgdG8gZ2x5Y2Vyb2wsIHdoaWNoIGlzIGF0dGFjaGVkIHRvIGEgcG9sYXIgJiM4MjIwO2hlYWQmIzgyMjE7IGNvbnNpc3Rpbmcgb2YgYSBwaG9zcGhhdGUgZ3JvdXAu
Cg==[Qq]
[q json=”true” multiple_choice=”true” xx=”2″ dataset_id=”SMV_biochem6_lipids_cumulative|8cd956e836b3b” question_number=”3″] The molecule shown below is a
[c]IGZhdHR5 IGFjaWQ=[Qq]
[f]IEV4Y2VsbGVudCEgVGhpcyBtb2xlY3VsZSBpcyBhIGZhdHR5IGFjaWQgKGFuIHVuc2F0dXJhdGVkIG9uZSku[Qq]
[c]IHRyaWdseWNlcmlkZQ==[Qq]
[f]IE5vLiBUcmlnbHljZXJpZGVzIGhhdmUgdGhyZWUgZmF0dHkgYWNpZHMgYXR0YWNoZWQgdG8gb25lIGdseWNlcm9sLg==[Qq]
[c]IHdheA==[Qq]
[f]IE5vLiBXYXhlcyBjb25zaXN0IG9mIHR3byBsb25nIGh5ZHJvY2FyYm9uIGNoYWlucywgYXQgbGVhc3Qgb25lIG9mIHdoaWNoIGlzIGEgZmF0dHkgYWNpZC4=
Cg==[Qq]
[c]IHN0ZXJvaWQ=[Qq]
[f]IE5vLiBTdGVyb2lkcyBjb25zaXN0IG9mIGZvdXIgZnVzZWQgY2FyYm9uIHJpbmdzLCBhcyBzaG93biBiZWxvdy4=
Cg==[Qq]
[c]IHBob3NwaG9saXBpZA==[Qq]
[f]IE5vLiBQaG9zcGhvbGlwaWRzIGNvbnNpc3Qgb2YgdHdvIG5vbi1wb2xhciBmYXR0eSBhY2lkIGNoYWlucywgYXR0YWNoZWQgdG8gZ2x5Y2Vyb2wsIHdoaWNoIGlzIGF0dGFjaGVkIHRvIGEgcG9sYXIgJiM4MjIwO2hlYWQmIzgyMjE7IGNvbnNpc3Rpbmcgb2YgYSBwaG9zcGhhdGUgZ3JvdXAu
Cg==[Qq]
[q json=”true” multiple_choice=”true” xx=”2″ dataset_id=”SMV_biochem6_lipids_cumulative|8cd4f951eb33b” question_number=”4″] The molecule shown below is a
[c]IGZhdHR5IGFjaWQ=[Qq]
[f]IE5vLiBUaGlzIG1vbGVjdWxlIGNvbnRhaW5zIGZhdHR5IGFjaWRzICh0aHJlZSBvZiB0aGVtLCBpbiBmYWN0KSwgYnV0IGl0JiM4MjE3O3Mgbm90IGp1c3QgYSBmYXR0eSBhY2lkLiBBIHNpbmdsZSBmYXR0eSBhY2lkIGlzIHNob3duIGJlbG93
Cg==[Qq]
[c]IHRyaWdseW NlcmlkZQ==[Qq]
[f]IE5pY2Ugam9iLiBUcmlnbHljZXJpZGVzIGhhdmUgdGhyZWUgZmF0dHkgYWNpZHMgYXR0YWNoZWQgdG8gb25lIGdseWNlcm9sLg==[Qq]
[c]IHdheA==[Qq]
[f]IE5vLiBXYXhlcyBjb25zaXN0IG9mIHR3byBsb25nIGh5ZHJvY2FyYm9uIGNoYWlucywgYXQgbGVhc3Qgb25lIG9mIHdoaWNoIGlzIGEgZmF0dHkgYWNpZC4=
Cg==[Qq]
[c]IHN0ZXJvaWQ=[Qq]
[f]IE5vLiBTdGVyb2lkcyBjb25zaXN0IG9mIGZvdXIgZnVzZWQgY2FyYm9uIHJpbmdzLCBhcyBzaG93biBiZWxvdy4=
Cg==[Qq]
[c]IHBob3NwaG9saXBpZA==[Qq]
[f]IE5vLiBQaG9zcGhvbGlwaWRzIGNvbnNpc3Qgb2YgdHdvIG5vbi1wb2xhciBmYXR0eSBhY2lkIGNoYWlucywgYXR0YWNoZWQgdG8gZ2x5Y2Vyb2wsIHdoaWNoIGlzIGF0dGFjaGVkIHRvIGEgcG9sYXIgJiM4MjIwO2hlYWQmIzgyMjE7IGNvbnNpc3Rpbmcgb2YgYSBwaG9zcGhhdGUgZ3JvdXAu
Cg==[Qq]
[q json=”true” multiple_choice=”true” xx=”2″ dataset_id=”SMV_biochem6_lipids_cumulative|8cd09bbb9fb3b” question_number=”5″] The molecule shown below is a
[c]IGZhdHR5IGFjaWQ=[Qq]
[f]IE5vLiBUaGlzIG1vbGVjdWxlIGNvbnRhaW5zIGZhdHR5IGFjaWRzICh0d28gb2YgdGhlbSwgaW4gZmFjdCksIGJ1dCBpdCYjODIxNztzIG5vdCBqdXN0IGEgZmF0dHkgYWNpZC4gQSBzaW5nbGUgZmF0dHkgYWNpZCBpcyBzaG93biBiZWxvdy4=
Cg==[Qq]
[c]IHRyaWdseWNlcmlkZQ==[Qq]
[f]IE5vLiBUcmlnbHljZXJpZGVzIGhhdmUgdGhyZWUgZmF0dHkgYWNpZHMgYXR0YWNoZWQgdG8gb25lIGdseWNlcm9sLg==[Qq]
[c]IHdheA==[Qq]
[f]IE5vLiBXYXhlcyBjb25zaXN0IG9mIHR3byBsb25nIGh5ZHJvY2FyYm9uIGNoYWlucywgYXQgbGVhc3Qgb25lIG9mIHdoaWNoIGlzIGEgZmF0dHkgYWNpZC4=
Cg==[Qq]
[c]IHN0ZXJvaWQ=[Qq]
[f]IE5vLiBTdGVyb2lkcyBjb25zaXN0IG9mIGZvdXIgZnVzZWQgY2FyYm9uIHJpbmdzLCBhcyBzaG93biBiZWxvdy4=
Cg==[Qq]
[c]IHBob3NwaG 9saXBpZA==[Qq]
[f]IEV4Y2VsbGVudC4gUGhvc3Bob2xpcGlkcyBjb25zaXN0IG9mIHR3byBub24tcG9sYXIgZmF0dHkgYWNpZCBjaGFpbnMsIGF0dGFjaGVkIHRvIGdseWNlcm9sLCB3aGljaCBpcyBhdHRhY2hlZCB0byBhIHBvbGFyICYjODIyMDtoZWFkJiM4MjIxOyBjb25zaXN0aW5nIG9mIGEgcGhvc3BoYXRlIGdyb3VwLg==[Qq]
[q json=”true” multiple_choice=”true” xx=”2″ dataset_id=”SMV_biochem6_lipids_cumulative|8ccc63661273b” question_number=”6″] A molecule that’s the key building block of cell membranes.
[c]IHN0ZXJvaWQ=[Qq]
[f]IE5vLiBXaGlsZSBjaG9sZXN0ZXJvbCBpcyBhIHN0ZXJvaWQgdGhhdCYjODIxNztzIGEgcGFydCBvZiBjZWxsIG1lbWJyYW5lcywgaXQmIzgyMTc7cyBub3QgdGhlIG1haW4gY29tcG9uZW50LiBZb3UmIzgyMTc7cmUgbG9va2luZyBmb3Igc29tZXRoaW5nIHRoYXQgb3JnYW5pemVzIGl0c2VsZiBpbnRvIGEgYmlsYXllci4=[Qq]
[c]IHRyaWdseWNlcmlkZQ==[Qq]
[f]IE5vLiBUcmlnbHljZXJpZGVzIGFyZSB1c2VkIGZvciBlbmVyZ3kgc3RvcmFnZSwgaW5zdWxhdGlvbiwgYW5kIGJ1b3lhbmN5LiBZb3UmIzgyMTc7cmUgbG9va2luZyBmb3Igc29tZXRoaW5nIHRoYXQgb3JnYW5pemVzIGl0c2VsZiBpbnRvIGEgYmlsYXllci4=[Qq]
[c]IHBob3NwaG 9saXBpZA==[Qq]
[f]IEV4Y2VsbGVudC4gUGhvc3Bob2xpcGlkcyBhcmUgdGhlIGtleSBidWlsZGluZyBibG9ja3Mgb2YgY2VsbCBtZW1icmFuZXMu[Qq]
[c]IHdheA==[Qq]
[f]IE5vLiBXYXhlcyBhcmUgdXNlZCBmb3Igd2F0ZXJwcm9vZmluZy4gWW91JiM4MjE3O3JlIGxvb2tpbmcgZm9yIGEgbW9sZWN1bGUgdGhhdCBvcmdhbml6ZXMgaXRzZWxmIGludG8gYSBiaWxheWVyLg==[Qq]
[q json=”true” multiple_choice=”true” xx=”2″ dataset_id=”SMV_biochem6_lipids_cumulative|8cc805cfc6f3b” question_number=”7″] A molecule that’s used for energy storage, insulation, and buoyancy.
[c]IHN0ZXJvaWQ=[Qq]
[f]IE5vLiBTdGVyb2lkcyBpbmNsdWRlIGhvcm1vbmVzIGxpa2UgdGVzdG9zdGVyb25lIGFuZCBlc3Ryb2dlbiwgYW5kIGFsc28gbW9sZWN1bGVzIGxpa2UgY2hvbGVzdGVyb2wsIGEga2V5IGNvbXBvbmVudCBvZiBjZWxsIG1lbWJyYW5lcy4=[Qq]
[c]IHRyaWdseW NlcmlkZQ==[Qq]
[f]IEV4Y2VsbGVudCEgVHJpZ2x5Y2VyaWRlcyBpbmNsdWRlIGZhdHMgYW5kIG9pbHMuIFRoZXkmIzgyMTc7cmUgdXNlZCBmb3IgZW5lcmd5IHN0b3JhZ2UsIGluc3VsYXRpb24sIGFuZCBidW95YW5jeS4=[Qq]
[c]IHBob3NwaG9saXBpZA==[Qq]
[f]IE5vLiBQaG9zcGhvbGlwaWRzIGFyZSB0aGUga2V5IGJ1aWxkaW5nIGJsb2NrcyBvZiBjZWxsIG1lbWJyYW5lcy4=[Qq]
[c]IHdheA==[Qq]
[f]IE5vLiBXYXhlcyBhcmUgdXNlZCBmb3Igd2F0ZXJwcm9vZmluZy4gWW91JiM4MjE3O3JlIGxvb2tpbmcgZm9yIHRoZSB0eXBlIG9mIG1vbGVjdWxlIHRoYXQgbWFrZXMgdXAgZmF0cyBhbmQgb2lscy4=[Qq]
[q json=”true” multiple_choice=”true” xx=”2″ dataset_id=”SMV_biochem6_lipids_cumulative|8cc35db7fef3b” question_number=”8″] The type of lipid that forms certain hormones, as well as the membrane component cholesterol.
[c]IHN0ZX JvaWQ=[Qq]
[f]IEF3ZXNvbWUhIFN0ZXJvaWRzIGluY2x1ZGUgaG9ybW9uZXMgbGlrZSB0ZXN0b3N0ZXJvbmUgYW5kIGVzdHJvZ2VuLCBhbmQgYWxzbyBtb2xlY3VsZXMgbGlrZSBjaG9sZXN0ZXJvbCwgYSBrZXkgY29tcG9uZW50IG9mIGNlbGwgbWVtYnJhbmVzLg==[Qq]
[c]IHRyaWdseWNlcmlkZQ==[Qq]
[f]IE5vLiBUcmlnbHljZXJpZGVzIGluY2x1ZGUgZmF0cyBhbmQgb2lscy4gVGhleSYjODIxNztyZSB1c2VkIGZvciBlbmVyZ3kgc3RvcmFnZSwgaW5zdWxhdGlvbiwgYW5kIGJ1b3lhbmN5Lg==[Qq]
[c]IHBob3NwaG9saXBpZA==[Qq]
[f]IE5vLiBQaG9zcGhvbGlwaWRzIGFyZSB0aGUga2V5IGJ1aWxkaW5nIGJsb2NrcyBvZiBjZWxsIG1lbWJyYW5lcy4=[Qq]
[c]IHdheA==[Qq]
[f]IE5vLiBXYXhlcyBhcmUgdXNlZCBmb3Igd2F0ZXJwcm9vZmluZy4=[Qq]
[q json=”true” multiple_choice=”true” xx=”2″ dataset_id=”SMV_biochem6_lipids_cumulative|8cbc619452f3b” question_number=”9″] A molecule used for waterproofing.
[c]IHN0ZXJvaWQ=[Qq]
[f]IE5vLiBTdGVyb2lkcyBpbmNsdWRlIGhvcm1vbmVzIGxpa2UgdGVzdG9zdGVyb25lIGFuZCBlc3Ryb2dlbiwgYW5kIGFsc28gbW9sZWN1bGVzIGxpa2UgY2hvbGVzdGVyb2wsIGEga2V5IGNvbXBvbmVudCBvZiBjZWxsIG1lbWJyYW5lcy4=[Qq]
[c]IHRyaWdseWNlcmlkZQ==[Qq]
[f]IE5vLiBUcmlnbHljZXJpZGVzIGluY2x1ZGUgZmF0cyBhbmQgb2lscy4gVGhleSYjODIxNztyZSB1c2VkIGZvciBlbmVyZ3kgc3RvcmFnZSwgaW5zdWxhdGlvbiwgYW5kIGJ1b3lhbmN5Lg==[Qq]
[c]IHBob3NwaG9saXBpZA==[Qq]
[f]IE5vLiBQaG9zcGhvbGlwaWRzIGFyZSB0aGUga2V5IGJ1aWxkaW5nIGJsb2NrcyBvZiBjZWxsIG1lbWJyYW5lcy4=[Qq]
[c]IHdh eA==[Qq]
[f]IEV4Y2VsbGVudC4gV2F4ZXMgYXJlIHVzZWQgZm9yIHdhdGVycHJvb2Zpbmcu[Qq]
[q]The molecules that make up that basic structure of the membrane are called [hangman].
[c]cGhvc3Bob2xpcGlkcw==[Qq]
[q]The two fatty acid chains make up the phospholipid’s “[hangman],” while the phosphate-containing part makes up the phospholipid’s “[hangman].”
[c]dGFpbA==[Qq]
[c]aGVhZA==[Qq]
[q]Whereas the head of a phospholipid is [hangman], the tail is [hangman].
[c]aHlkcm9waGlsaWM=[Qq]
[c]aHlkcm9waG9iaWM=[Qq]
[q]The non-[hangman] tails of a phospholipid won’t form [hangman] bonds with water molecules. That’s why they’re [hangman].
[c]cG9sYXI=[Qq]
[c]aHlkcm9nZW4=[Qq]
[c]aHlkcm9waG9iaWM=[Qq]
[q]The arrangement of phospholipids shown below is called a phospholipid [hangman].
[c]YmlsYXllcg==[Qq]
[x][restart][/qwiz]
What’s next?
- Continue to AP Bio Topic 1.7, Proteins (the next tutorial in Unit 1. We’re doing topic 1.7 (proteins) before 1.6 (nucleic acids)).