Looking for a student learning guide? It’s on the high school main menu.
1. Why the molecules of life are built around carbon

By weight, living things are mostly made out of water. As an example, just look in the mirror. According to the US Geological Survey, your body is about 60% water by weight. For a tree, the percentage of weight composed of water can be more than 2/3 (Source: University of Illinois).
But while water makes up most of the weight of living things, the shape, structure, and function of living things comes from the element carbon. Life is astoundingly complex. Carbon is the basis of that complexity. Here’s why.
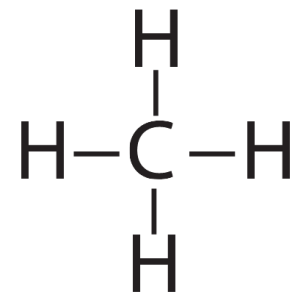
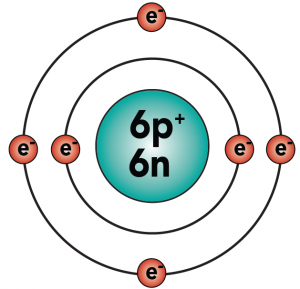
Carbon is element number six in the periodic table. It’s number six because it has six protons, along with six neutrons and six electrons.
With four outer (also known as valence) electrons, carbon readily forms covalent bonds with other atoms, including other carbon atoms.
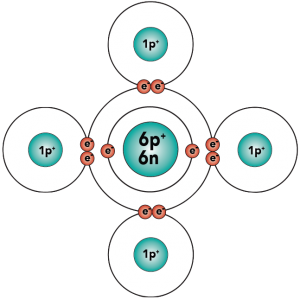 |
|
|
Electron distribution model |
Structural formula |
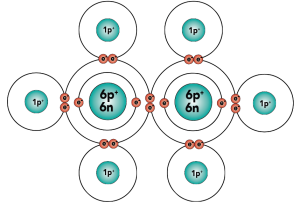 |
 |
| Electron distribution model | Structural formula |
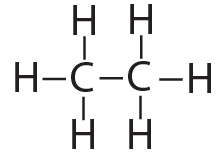
That carbon-carbon sharing makes it possible for carbon to form chains, rings, and branched molecules.
| Chain | Branched molecules | Rings |
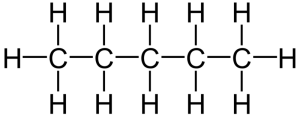 |
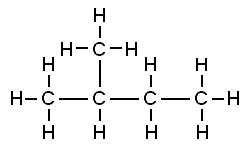 |
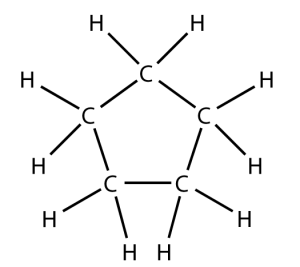 |
This complexity and variety is further enhanced by carbon’s ability to form double bonds and triple bonds, as shown below…
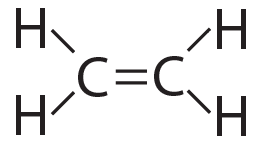
Ethylene (CH4) has a double bond |
Acetylene (CH2) has a triple bond |
So why is this important? Living things do phenomenally complex things, like
- Taking in matter and energy from the environment to grow and develop,
- Reproducing and passing on their genes to the next generation.
- Moving parts of themselves or their entire bodies from one location to another.
The ability of carbon to form bonds with other atoms, particularly with itself, makes the large, complex molecules that carry out these activities possible.
Below, we’ll start seeing how the molecules of life are constructed. But before that, let’s check to make sure that you’re clear on what’s above.
2. Checking Understanding: Carbon: Life’s Essential Element
[qwiz qrecord_id=”sciencemusicvideosMeister1961-Why Carbon (HS)”]
[h]Carbon: Life’s Essential Element
[i]
[q]Carbon has _____ protons.
[textentry single_char=”true”]
[c]Ng ==[Qq]
[f]WWVzLiBDYXJib24gaGFzIDYgcHJvdG9ucy4=[Qq]
[c]Kg==[Qq]
[f]Tm8uIEJ1dCB5b3UgY2FuIGZpZ3VyZSBpdCBvdXQgYnkgZXhhbWluaW5nIHRoaXMgZGlhZ3JhbS4=[Qq]
[q]Carbon has _______ outer (or valence) electrons.
[textentry single_char=”true”]
[c]NA ==[Qq]
[f]WWVzLiBDYXJib24gaGFzIDTCoG91dGVyIChvciB2YWxlbmNlKSBlbGVjdHJvbnMu[Qq]
[c]Kg==[Qq]
[f]Tm8uIEJ1dCB5b3UgY2FuIGZpZ3VyZSBpdCBvdXQgYnkgZXhhbWluaW5nIHRoaXMgZGlhZ3JhbS4=
Cg==[Qq]
[q]In ethane, is the bond connecting the two carbons a single bond or a double bond?
[c]c2lu Z2xl[Qq]
[c]ZG91Ymxl[Qq]
[f]Q29ycmVjdC4gVGhlIGJvbmQgYmV0d2VlbiB0aGUgdHdvIGNhcmJvbnMgKGFuZCBiZXR3ZWVuIHRoZSBjYXJib25zIGFuZCB0aGUgaHlkcm9nZW5zKSBpcyBhIHNpbmdsZSBib25kLCBpbnZvbHZpbmcgYSBzaW5nbGUgc2hhcmVkIHBhaXIgb2YgZWxlY3Ryb25zLg==[Qq]
[f]Tm8uIFdoZW4gYXRvbXMgc2hhcmUgYSBzaW5nbGUgcGFpciBvZiBlbGVjdHJvbnMsIGl0JiM4MjE3O3MgYSBzaW5nbGUgYm9uZC4=[Qq]
[q]Because carbon has four outer electrons, it can be organized into a variety of forms. Label these forms in the table below.
| ______________ | _____________ | ____________ |
[l]branches
[fx] No. Please try again.
[f*] Good!
[l]chains
[fx] No. Please try again.
[f*] Great!
[l]rings
[fx] No. Please try again.
[f*] Excellent!
[q]By weight, living things are mostly composed of [hangman]
[c]d2F0ZXI=[Qq]
[q]While water makes up most of the mass of living things, the element that’s the basis for life’s complex chemistry is the element [hangman].
[c]Y2FyYm9u[Qq]
[/qwiz]
3. The Four Families of Biological Molecules
Here’s a challenge for your imagination.
Think of the four most different living things that you can imagine.
I think of the 100 meter tall California redwoods, the 2 millionth of a meter E.coli bacteria that live in our large intestines, the fungi that live in association with plant roots, and Orcas (also known as killer whales).
These organisms, despite their different forms, sizes, and ecological niches, are built from the same four types of molecules. Ignoring the water which makes up most of the mass of any organism, they’re each composed of carbohydrates, lipids, proteins, and nucleic acids.

These groups of molecules are called biomolecule families. “Biomolecule” simple means “molecules in living things.” Except for the nucleic acids, these biomolecule families are probably familiar to you, because they’re also food groups. In this context, you might not have heard of lipids, but you eat them all the time. They include the yummy and energy-rich fats and oils.
You can see three biomolecule families listed in the nutrition facts for the potato chips on the right. Find the fat (a lipid), the carbohydrate, and the protein.
To connect this section with the previous one, these biomolecule families are the complex carbon-based molecules that make up living things. To start learning about them, take a moment and study the list below.
- Carbohydrates include
- sugars, which cells use for energy
- starches, which plants use for energy storage
- plant fiber (also known as cellulose), which plants use to build their cell walls, as well as woody stems, and other structures.
- Lipids include
- Fats and oils for energy storage
- Phospholipids, which make up cell membranes
- Waxes, for water proofing
- Hormones that regulate body functions and influence development.
- Proteins include
- Muscle tissue that allows your body to move. Most of the meat we eat is muscle tissue.
- Structural proteins that make up skin, hair, and fingernails;
- Enzymes, which control the chemical reactions in all living things, and
- Antibodies (immune system molecules that fight infections from bacteria and viruses).
- Nucleic Acids include
- DNA, the molecule that makes up genes;
- RNA, a molecule used to direct cellular activities; and
- ATP, the molecule used by cells to meet their moment to moment energy needs.
Note that the term biomolecule is roughly synonymous with the term macromolecule. The prefix “macro” refers to the fact that these molecules are big, consisting of anywhere from a dozen to thousands of atoms.
Let’s consolidate what’s in the list above with a quiz.
4. Checking Understanding: The Four Biomolecule Families
[qwiz qrecord_id=”sciencemusicvideosMeister1961-Four Biomolecule Families (HS)”]
[q labels = “top”]
| Family | Carbohydrates | Lipids | Proteins | Nucleic Acids |
| Example | _________ | __________ | _________ | _________ |
| Function | _________ _________ |
__________ __________ |
___________ ___________ |
_________ _________ |
[l]DNA
[f*] Correct!
[fx] No, that’s not correct. Please try again.
[l]energy storage
[f*] Great!
[fx] No. Please try again.
[l]energy source
[f*] Excellent!
[fx] No. Please try again.
[l]fat
[f*] Correct!
[fx] No. Please try again.
[l]genetic info
[f*] Excellent!
[fx] No. Please try again.
[l]movement
[f*] Good!
[fx] No. Please try again.
[l]muscle
[f*] Correct!
[fx] No. Please try again.
[l]sugar
[f*] Good!
[fx] No, that’s not correct. Please try again.
[q multiple_choice=”true”] The molecules of heredity are
[c]IGxpcGlkcw==[Qq]
[f]IE5vLiBMaXBpZHMgYXJlIGNvbm5lY3RlZCB3aXRoIGVuZXJneSBzdG9yYWdlIChmYXRzIGFuZCBvaWxzKSwgY2VsbCBtZW1icmFuZXMgKHBob3NwaG9saXBpZHMpLCB3YXRlcnByb29maW5nICh3YXhlcyksIGFuZCBjZXJ0YWluIGhvcm1vbmVzLg==[Qq]
[c]IGNhcmJvaHlkcmF0ZXM=[Qq]
[f]IE5vLiBDYXJib2h5ZHJhdGVzIGluY2x1ZGUgZW5lcmd5IHNvdXJjZXMgbGlrZSBzdWdhcnMsIGVuZXJneSBzdG9yYWdlIG1vbGVjdWxlcyBsaWtlIHN0YXJjaCwgYW5kIHBsYW50IGZpYmVycy4=[Qq]
[c]IHByb3RlaW5z[Qq]
[f]IE5vLiBQcm90ZWlucyBhcmUgdGhlIGFjdGlvbiBtb2xlY3VsZXMgb2YgdGhlIGNlbGwsIGFsbG93aW5nIGZvciBkZWZlbnNlLCBtb3ZlbWVudCwgYW5kIGVuenltYXRpYyBjb250cm9sIG9mIHRoZSBjZWxsJiM4MjE3O3MgY2hlbWlzdHJ5[Qq]
[c]IG51Y2xlaW MgYWNpZHM=[Qq]
[f]IEV4Y2VsbGVudC4gTnVjbGVpYyBhY2lkcyBsaWtlIEROQSBhcmUgdGhlIG1vbGVjdWxlcyBvZiBoZXJlZGl0eS4=[Qq]
[q multiple_choice=”true”] Functions like providing cells with energy, creating plant fibers, and some long term energy storage in the form of starch are associated with
[c]IGxpcGlkcw==[Qq]
[f]IE5vLiBMaXBpZHMgYXJlIGNvbm5lY3RlZCB3aXRoIGVuZXJneSBzdG9yYWdlIChmYXRzIGFuZCBvaWxzKSwgY2VsbCBtZW1icmFuZXMgKHBob3NwaG9saXBpZHMpLCB3YXRlcnByb29maW5nICh3YXhlcyksIGFuZCBjZXJ0YWluIGhvcm1vbmVzLg==[Qq]
[c]IGNhcmJvaH lkcmF0ZXM=[Qq]
[f]IE5pY2Ugam9iLiBDYXJib2h5ZHJhdGVzIGluY2x1ZGUgZW5lcmd5IHNvdXJjZXMgbGlrZSBzdWdhcnMsIGVuZXJneSBzdG9yYWdlIG1vbGVjdWxlcyBsaWtlIHN0YXJjaCwgYW5kIHBsYW50IGZpYmVycy4=[Qq]
[c]IHByb3RlaW5z[Qq]
[f]IE5vLiBQcm90ZWlucyBhcmUgdGhlIGFjdGlvbiBtb2xlY3VsZXMgb2YgdGhlIGNlbGwsIGFsbG93aW5nIGZvciBkZWZlbnNlLCBtb3ZlbWVudCwgYW5kIGVuenltYXRpYyBjb250cm9sIG9mIHRoZSBjZWxsJiM4MjE3O3MgY2hlbWlzdHJ5[Qq]
[c]IG51Y2xlaWMgYWNpZHM=[Qq]
[f]IEV4Y2VsbGVudC4gTnVjbGVpYyBhY2lkcyBsaWtlIEROQSBhcmUgdGhlIG1vbGVjdWxlcyBvZiBoZXJlZGl0eS4=[Qq]
[q multiple_choice=”true”] The action molecules of the cell, involved with functions such as defense, controlling chemical reactions, and movement are
[c]IGxpcGlkcw==[Qq]
[f]IE5vLiBMaXBpZHMgYXJlIGNvbm5lY3RlZCB3aXRoIGVuZXJneSBzdG9yYWdlIChmYXRzIGFuZCBvaWxzKSwgY2VsbCBtZW1icmFuZXMgKHBob3NwaG9saXBpZHMpLCB3YXRlcnByb29maW5nICh3YXhlcyksIGFuZCBjZXJ0YWluIGhvcm1vbmVzLg==[Qq]
[c]IGNhcmJvaHlkcmF0ZXM=[Qq]
[f]IE5vLiBDYXJib2h5ZHJhdGVzIGluY2x1ZGUgZW5lcmd5IHNvdXJjZXMgbGlrZSBzdWdhcnMsIGVuZXJneSBzdG9yYWdlIG1vbGVjdWxlcyBsaWtlIHN0YXJjaCwgYW5kIHBsYW50IGZpYmVycy4=[Qq]
[c]IHByb3 RlaW5z[Qq]
[f]IEZhYnVsb3VzLiBQcm90ZWlucyBhcmUgdGhlIGFjdGlvbiBtb2xlY3VsZXMgb2YgdGhlIGNlbGwsIGFsbG93aW5nIGZvciBkZWZlbnNlLCBtb3ZlbWVudCwgYW5kIGVuenltYXRpYyBjb250cm9sIG9mIHRoZSBjZWxsJiM4MjE3O3MgY2hlbWlzdHJ5[Qq]
[c]IG51Y2xlaWMgYWNpZHM=[Qq]
[f]IE5vLiBOdWNsZWljIGFjaWRzIGxpa2UgRE5BIGFyZSB0aGUgbW9sZWN1bGVzIG9mIGhlcmVkaXR5Lg==[Qq]
[q multiple_choice=”true”] Fats and oils, waxes, certain hormones, and the molecules that make up cell membranes are all part of which biomolecule family?
[c]IGxpcG lkcw==[Qq]
[f]IFdheSB0byBnbyEgTGlwaWRzIGFyZSBjb25uZWN0ZWQgd2l0aCBlbmVyZ3kgc3RvcmFnZSAoZmF0cyBhbmQgb2lscyksIGNlbGwgbWVtYnJhbmVzIChwaG9zcGhvbGlwaWRzKSwgd2F0ZXJwcm9vZmluZyAod2F4ZXMpLCBhbmQgY2VydGFpbiBob3Jtb25lcy4=[Qq]
[c]IGNhcmJvaHlkcmF0ZXM=[Qq]
[f]IE5vLiBDYXJib2h5ZHJhdGVzIGluY2x1ZGUgZW5lcmd5IHNvdXJjZXMgbGlrZSBzdWdhcnMsIGVuZXJneSBzdG9yYWdlIG1vbGVjdWxlcyBsaWtlIHN0YXJjaCwgYW5kIHBsYW50IGZpYmVycy4=[Qq]
[c]IHByb3RlaW5z[Qq]
[f]IE5vLiBQcm90ZWlucyBhcmUgdGhlIGFjdGlvbiBtb2xlY3VsZXMgb2YgdGhlIGNlbGwsIGFsbG93aW5nIGZvciBkZWZlbnNlLCBtb3ZlbWVudCwgYW5kIGVuenltYXRpYyBjb250cm9sIG9mIHRoZSBjZWxsJiM4MjE3O3MgY2hlbWlzdHJ5Lg==[Qq]
[c]IG51Y2xlaWMgYWNpZHM=[Qq]
[f]IE5vLiBOdWNsZWljIGFjaWRzIGxpa2UgRE5BIGFyZSB0aGUgbW9sZWN1bGVzIG9mIGhlcmVkaXR5Lg==[Qq]
[/qwiz]
5. Monomers and Polymers
We’ll get to know the important details about each of the biomolecule families later in this set of tutorials. But now let’s turn to a related point. Cells build most biomolecules (carbohydrates, proteins, and nucleic acids) by piecing together molecular building blocks called monomers. Note the prefix “mono,” which indicates “one” (as in monorail, monotheism, monosyllabic, etc.). Monomers are groups of atoms that function as a unit.
 To get a sense of how monomers are used, let’s think about Legos. Legos are composed of a plastic called acrylonitrile butadiene styrene (ABS). But children around the world who play with Legos don’t need to know that or think about it. They just combine individual Lego bricks into larger structures, with new attributes. One Lego brick is just a brick. A few hundred can be the Millenium Falcon.
To get a sense of how monomers are used, let’s think about Legos. Legos are composed of a plastic called acrylonitrile butadiene styrene (ABS). But children around the world who play with Legos don’t need to know that or think about it. They just combine individual Lego bricks into larger structures, with new attributes. One Lego brick is just a brick. A few hundred can be the Millenium Falcon.
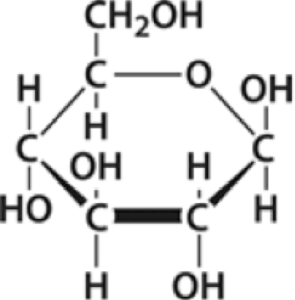
In biological systems, cells use monomers in a similar way. For example, the molecule at left is glucose (C6H12O6) . Glucose is one of the monomers of carbohydrates. It’s a simple sugar that’s made by plants during photosynthesis, and which is used as an energy source by both plants and animals.
As you can see, glucose is made of carbon, hydrogen, and oxygen atoms. When cells need to build something out of glucose, they use glucose as a unit. For example, if plant cells need to store chemical energy, they’ll combine glucose molecules together to form starch.
As with our Legos, the monomer and what it gets made into have different properties. Glucose is sweet. As you know from the starchy foods you eat (bread, pasta rice, potatoes), starch isn’t sweet. Glucose easily dissolves in water. Starch doesn’t easily dissolve.
Take a moment and study the diagram of starch below. In this diagram, the glucose monomers are drawn slightly differently from what’s above, but you should be able to see that starch consists of a chain of glucose molecules. Also, note that while 9 combined glucose monomers are shown in the starch molecule below, an actual starch molecule could consist of hundreds of glucose subunits.
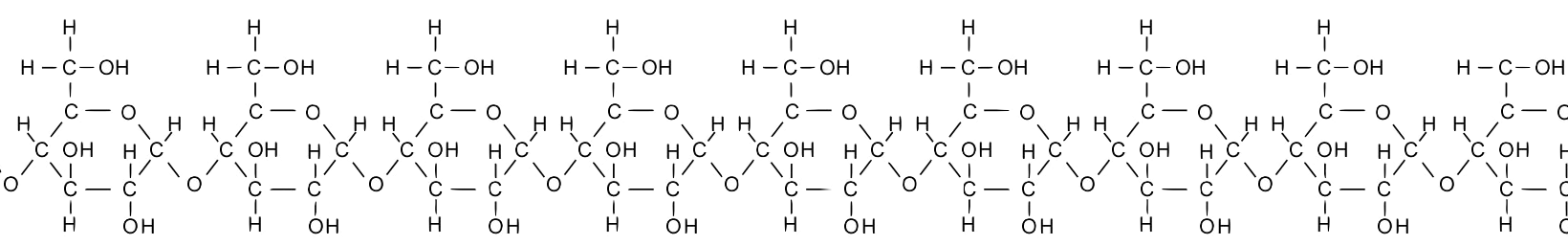
Chaining monomers together, as in the example of starch above, creates polymers, larger molecules that are built of two or more chemically combined monomers. The prefix “poly” means “many,” as in polytheism or polygon.
In addition to having different properties from their monomers, polymers are much more diverse than monomers. A useful analogy is to think of letters and words, with letters as the monomers and words as the polymers. English has 26 letters. We combine letters to form words. According to an article in the Oxford Living Dictionary, English has over 170,000 words (with other sources citing more).
You can see this explosion of polymer diversity by considering proteins, a class of biological polymers that make up muscle, skin, enzymes and many other biological structures. The monomers of proteins are amino acids, of which there are twenty. While there are twenty amino acids, nobody knows how many proteins exist in the living world. In the human body alone there might be as many as three million distinct proteins. And while many of the proteins found in other species are the same as those found in humans, many aren’t. It’s a great example of the biological theme of unity and diversity. We’re all made of the same building blocks (unity). But the structures built from those building blocks can have practically infinite variety (diversity).
Here’s one more thought about the “monomers are letters/polymers are words” analogy. In English, the set of 170,000 words (using the low estimate) that’s generated from our 26 letter alphabet is made with a few constraints. For example, it’s unusual, in English, for words to have more than three consonants in a row. As you might have noticed in the discussion of starch above, there’s no parallel to that when combining monomers together. Starch can be hundreds of glucose monomers in a row. Also, the average length of a word in English is about 5 letters. There are only 1435 words that are 15 letters long, and 401 words that are 17 letters long. Source:YouGoWords! In living things, monomer combinations are just getting started at that length. Hemoglobin, the oxygen carrying protein in your red blood cells, is composed of polymer chains that are about 140 monomers long. The enzyme rubisco, the protein partly responsible for making carbon dioxide into sugars during photosynthesis, has about 500 amino acids.Source: CellBiologybytheNumbers.
6. Dehydration synthesis is how monomers become polymers
In living cells, combining monomers into polymers occurs through a process called dehydration synthesis. The synthesis part of that term is straightforward: to synthesize means “to put together,” which is what’s happening when monomers become polymers. It’s called dehydration synthesis because a water molecule is removed from the monomers that are being combined together. Who’s combining them? We’ll study this much more in a later module, but you should know for now that when monomers are combined into polymers, the “agents” doing the combining are proteins called enzymes.
Here’s a visual representation of a dehydration synthesis reaction.
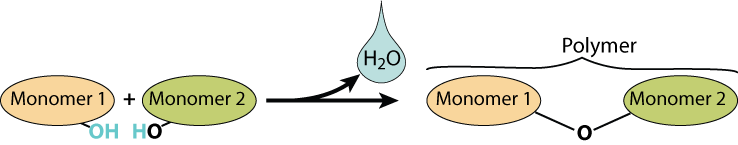
An enzyme (not shown) combines the two monomers. As it does, a water molecule is created as an —OH is removed from the monomer on the left and an —H is removed from the monomer on the right. The H— and the —OH combine to form H2O (water)
Here’s another look at a dehydration synthesis reaction.
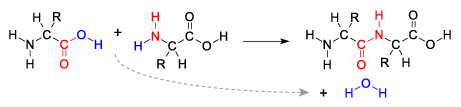
The two reacting molecules are amino acids, the monomers of proteins that we discussed above. Just as in the cartoon version of a dehydration synthesis above, note that on the right side of the first amino acid is an —OH, and that on the left side of the second amino acid is an —H (a hydrogen atom). Both of these are colored blue to make it easy for you to find them. As these two amino acids are linked together, a water molecule (also in blue) is removed.
 Note that once that bond between the two monomers is formed, it’s just as strong as any other bond within the molecule. It’s not as if the monomers are Legos that have been easily snapped together, and the polymers are just as easily snapped apart. The bond between the red colored carbon and nitrogen in the two linked amino acids above is a full-fledged single covalent bond. The polymer, chemically speaking, is one molecule.
Note that once that bond between the two monomers is formed, it’s just as strong as any other bond within the molecule. It’s not as if the monomers are Legos that have been easily snapped together, and the polymers are just as easily snapped apart. The bond between the red colored carbon and nitrogen in the two linked amino acids above is a full-fledged single covalent bond. The polymer, chemically speaking, is one molecule.
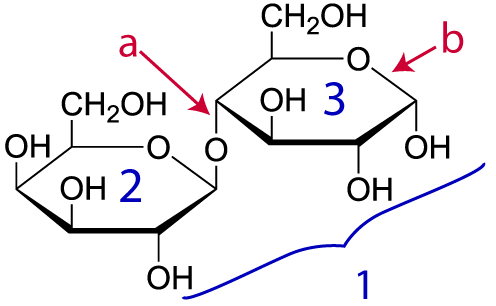
Here’s another example. Take a look at lactose (“1,” at right). Lactose is the sugar in milk. Don’t panic about how different this structural formula looks from the ones above. A convention among biochemists is to let you assume that there’s a carbon and attached hydrogen atoms at every place you see an angle vertex. So, lactose has the molecular formula C12H22O11, even though only two carbons, nine hydrogens, and ten oxygens are explicitly shown in the diagram.
Lactose is composed of two monomers: “2” is the simple sugar galactose. “3” is glucose (which we met above). Letter “a” is the bond holding these monomers together. That bond is a single covalent bond (which, you should remember, involves a shared pair of electrons: click here for a review). It’s just as strong as any other bond, such as the bond at “b.”
That having been said, while there are enzymes that attach monomers together to make polymers, there are other enzymes that pull polymers apart, breaking them into their constituent monomers. When that happens, the enzymes keep the monomers’ integrity intact. In other words, there’s an enzyme (called lactase) that breaks lactose apart into the monomers galactose and glucose, and it does that by breaking the bond at “a” above. Let’s see how.
7. Hydrolysis is how polymers become monomers
The opposite of dehydration synthesis is hydrolysis. The hydro part of that term refers to water. The lysis part refers to splitting or breaking, and you’ll see lysis comes up as both a term on its own and as a part of other biology terms throughout this course. So hydrolysis means splitting with water. Enzymes pull out a monomer by “feeling” where the monomers are connected, and jamming a water molecule in between them, causing the bond between the monomers to break. In cartoon form, it looks like this:
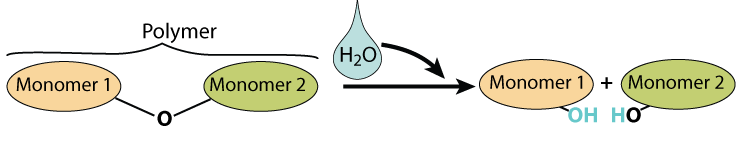
And here’s what hydrolysis looks like with structural formulas, using lactose as an example.
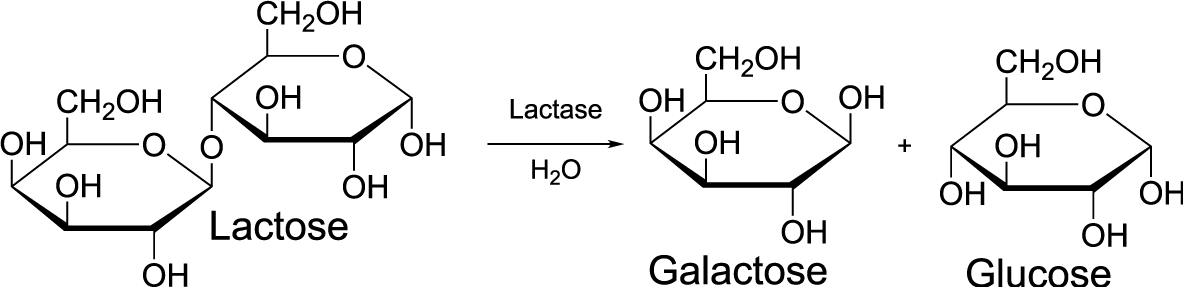
Let’s think about the hydrolysis that’s taking place. Over the arrow in between the monomer and the polymer you see the word “lactase:” this is the enzyme that takes lactose and breaks it apart into galactose (the simple sugar on the left) and glucose (the simple sugar on the right). Notice that under the arrow you see H2O. That represents the water molecule that gets jammed in between the lactose monomers as the bond between them is broken. You could also write out a word equation for the hydrolysis of lactose as follows:
lactose + water → galactose + glucose
5. Quiz: Monomers, Polymers, and the Four Families of Biological Molecules
[qwiz use_dataset=”SMV_biochem1_Monomers and Polymers” dataset_intro=”false” random = “false” qrecord_id=”sciencemusicvideosMeister1961-Monomers and polymers (HS)”] [h]
Biochemistry Basics
[i]
[x][restart]
[/qwiz]
Links
- Carbohydrates (the next interactive tutorial in this module)
- The Molecules of Life (High School Level): Main Menu