Page Outline
- The Functions of Proteins
- Proteins are Polymers of Amino Acids, Linked by Peptide Bonds
- Amino Acids, Peptide Bonds, and Polypeptide Quiz
- Protein structure
- Denaturation
- Proteins quiz
1. The Functions of Proteins
Let’s start our study of proteins by considering our hands.

Our fingernails are composed of a protein called keratin. A different form of keratin makes up the outermost layer of your skin. The elasticity of deeper layers of your skin — your skin’s ability to return to its shape after being pinched or stretched — is made possible by a protein called elastin.
Go a bit deeper. Your fingers move because they’re attached to muscles by tendons and ligaments that are composed of a protein called collagen. The muscles that move the bones in your hand are made of proteins such as actin and myosin.
Think about your veins. The red blood cells that flow within them are filled with an oxygen-carrying protein called hemoglobin. If you were to cut your hand and get an infection, your immune system would produce proteins called immunoglobulins to form the antibodies that would beat back the invading bacteria.
When you look at any animal, you’re looking at protein. The same is not true of plants, which are mostly carbohydrate. But how did that carbohydrate get there? The enzyme that pulls carbon dioxide out of the air to create carbohydrates during photosynthesis is called Rubisco. Like almost all enzymes, it’s a protein.
I could go on. But let’s organize our thinking about the various functions of proteins by completing the interactive table below. Use prior knowledge and trial and error (and don’t worry about getting every answer right the first time).
[qwiz qrecord_id=”sciencemusicvideosMeister1961-Biochem: Functions of Proteins Table”]
[h]Some of the functions of proteins
[q labels = “top”]
Proteins
- Control chemical reactions as ______________.
- Create _____________ like bone, hair, feathers (keratin)
- Fight __________ (antibodies)
- Produce movement (___________)
- ____________ oxygen (hemoglobin in red blood cells)
- Store __________________ (albumin in egg white)
- Transmit _________ as hormones and neurotransmitters.
PROTEINS ARE THE ___________________ MACROMOLECULES
[l]chemical energy
[fx] No, that’s not correct. Please try again.
[f*] Excellent!
[l]infection
[fx] No. Please try again.
[f*] Correct!
[l]enzymes
[fx] No, that’s not correct. Please try again.
[f*] Excellent!
[l]MOST DIVERSE
[fx] No, that’s not correct. Please try again.
[f*] Great!
[l]muscle
[fx] No. Please try again.
[f*] Correct!
[l]structures
[fx] No. Please try again.
[f*] Excellent!
[l]transport
[fx] No. Please try again.
[f*] Correct!
[l]signals
[fx] No. Please try again.
[f*] Good!
[/qwiz]
2. Proteins are polymers of amino acids, linked by peptide bonds
2a. Amino Acids
We’ve learned previously that polysaccharides are polymers of monosaccharides and that nucleic acids are polymers of nucleotides. Similarly, proteins are polymers of amino acids.
There are twenty amino acids found in living things. Each one has a three-letter abbreviation: “lys” for lysine, “ser” for serine, etc. All are variations of a common structure.
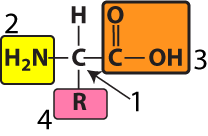
At the center of every amino acid is a central carbon atom (at “1” in the diagram to your left). The central carbon is also referred to as the alpha (α) carbon. This central carbon is flanked by an amino group (“2”) and a carboxylic acid (at “3”). That’s where the name “amino acid” comes from.
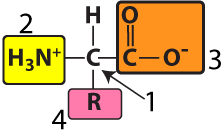
Note that the amino group and the carboxyl group can be shown in both their non-ionized (as shown on the left) and their ionized forms (as shown on the right). You should be able to recognize both.
At “4” is an “R group” or “side chain.” Each of the twenty amino acids has a distinct R group. The R groups are shaded in the diagram below.
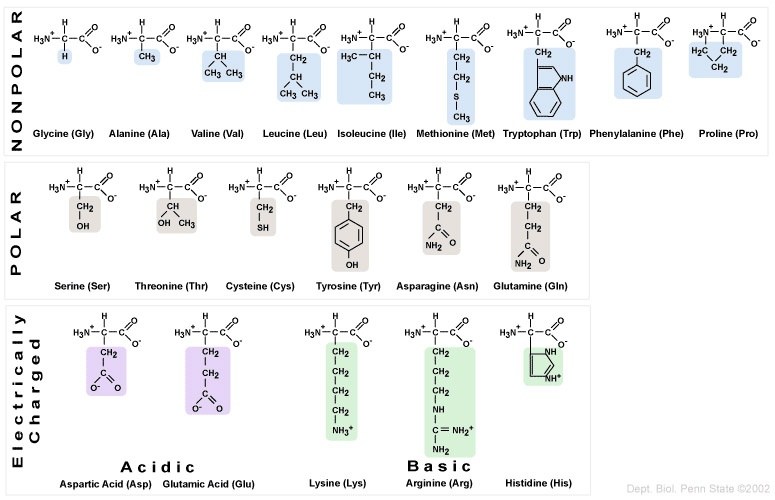
The twenty amino acids can be organized into four groups, based on the chemistry of their side chains. These R groups can be
- Non-polar/hydrophobic
- Polar/hydrophilic
- Acidic (leaving them with a negative charge after they’ve donated a proton to the solution)
- Basic (leaving them with a positive charge after they’ve absorbed a proton from the solution)
Following a few rules, you can easily identify which category an amino acid falls into.
- Non-polar: R groups with methyl functional groups (—CH3), or with fused carbon rings.
- Polar: R groups with hydroxyl (—OH) or sulfhydryl (—SH) functional groups.
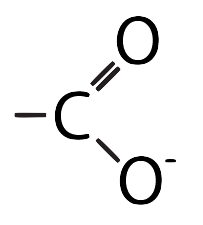
- Acidic: R groups with carboxylic acid functional groups.
- Basic: R groups with amino functional groups: NH3+ or NH2+ or NH.
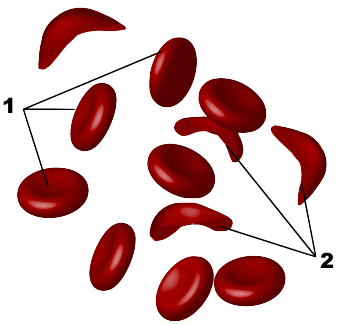
This is important because mutations in DNA can result in substitutions of one amino acid for another. If the substituted amino acid has a different chemistry than the original amino acid, the effect on the protein’s structure and function can be significant. For example, the inherited blood disorder sickle cell anemia is caused by a substitution of glutamic acid for valine in a protein called hemoglobin, which carries oxygen in red blood cells. Glutamic acid is (obviously) an acid, while valine is non-polar. The substitution changes hemoglobin’s chemistry so that it polymerizes under low oxygen conditions, causing red blood cells to become spiked (“2”) instead of smooth (“1”), blocking capillaries and causing tissue damage.
2.b. Peptide Bonds
During the process of protein synthesis, ribosomes (tiny particles within cells, represented by “2” below) link amino acids (“1”) together, following instructions sent to them from the cell’s DNA. The instructions are in the form of a single-stranded nucleic acid called messenger RNA, shown at “3” below.
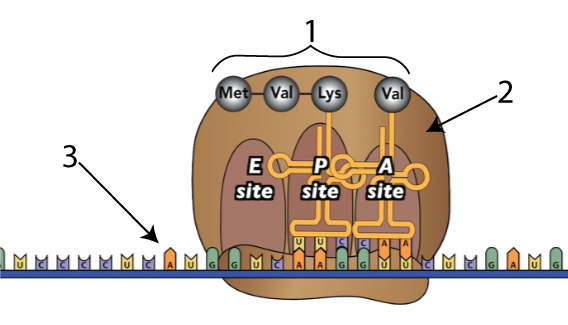
Here’s what happens as ribosomes link amino acids together.
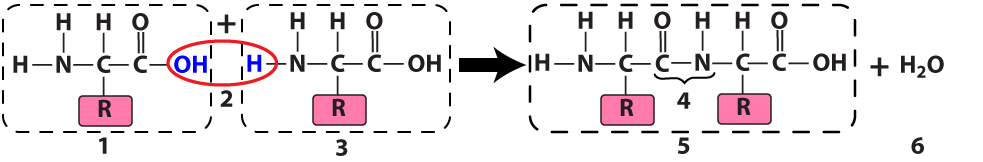
The ribosome connects two amino acids (“1” and “3”) by removing a hydroxyl functional group (an —OH) from one amino acid and a hydrogen atom from another. The result is a dipeptide (“5”): two amino acids linked by a peptide bond (at “4”). Because this is a dehydration synthesis, a water molecule (at “6”) also results from this reaction.
2c. Polypeptides
Add more amino acids and you have a polypeptide (a chain of linked amino acids).
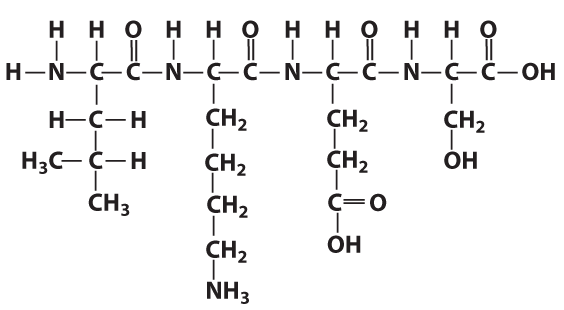
In the same way as the letters in a word can be in any order, the amino acids in a protein can be in any order. As I’ve noted previously, however, the “letters to words = amino acids to protein” analogy is limited. Proteins can be thousands of amino acids long, making the universe of potential proteins much greater than the universe of potential words.
Let’s end this section with a bit of protein terminology (all of which has shown up on AP bio exams since the redesign in 2012).
- Each of the ends of a protein/polypeptide chain has a specific name. The amino acid at the start of a protein has an exposed amino functional group (an —NH2). That amino acid is called the N terminal amino acid, and that end is called the N terminus. The amino acid at the other end of a protein has an exposed carboxyl group (—COOH), making that the C-terminal amino acid, and that end of the C-terminus. You’ll see these terms come up in relationship to protein-digesting enzymes, some of which can only break off amino acids at the N terminus, while others are limited to the C terminus.
- An amino acid within a polypeptide chain is sometimes referred to as a residue (because it’s what’s left over after the dehydration synthesis reaction that put it there). For example, in a discussion of the mutation in hemoglobin that causes sickle cell anemia, you might read that “the mutation is in the sixth amino acid residue,” as shown below
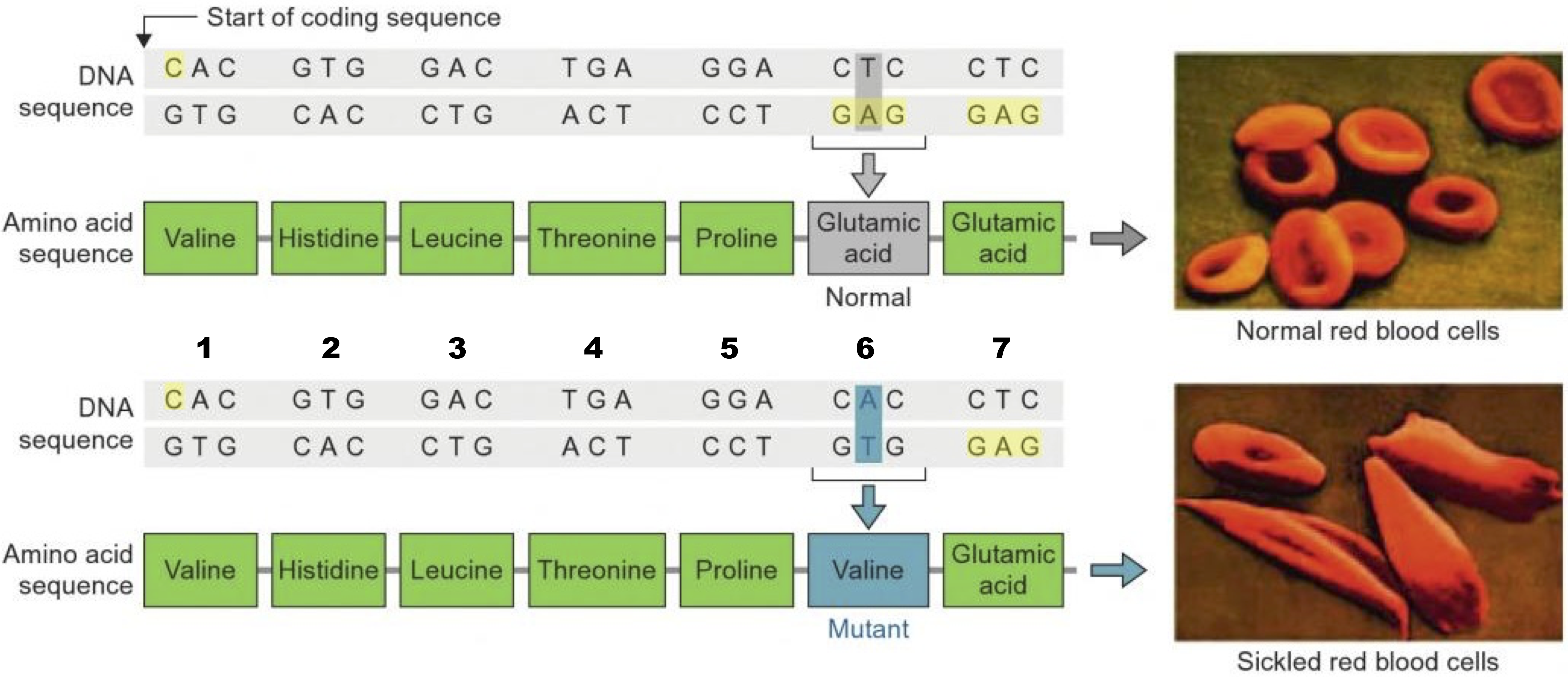
They’re just saying that it’s in the sixth amino acid of the chain (and now it won’t bother you when you see it!).
3. Amino Acids, Peptide Bonds, and Polypeptides: Checking Understanding
[qwiz use_dataset=”SMV_biochem_7_amino acids and peptide bonds” dataset_intro=”false” qrecord_id=”sciencemusicvideosMeister1961-SMV-peptide bonds and amino acids (v2)”] [h]
Proteins Quiz 1
[i]
[x][restart][/qwiz]
4. Protein Structure
4a. Proteins are Specific and Dynamic
Protein structure has a few remarkable features. The first is specificity of shape. To get an understanding of this, let’s think about our immune system.
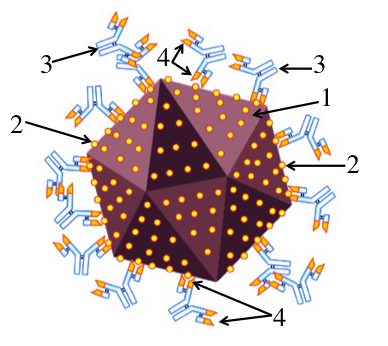
Imagine that you’ve been infected by a virus (“1”). Your immune system counterattacks by producing proteins called antibodies (“3”) that can bind with specific molecules on the surface of the virus. These molecules are called antigens (“2”).
An antibody can bind with an antigen because the tip of the antibody has an antigen-binding site (“4”) whose shape closely matches with, or complements, the shape of the antigen. This binding either directly neutralizes the virus (keeping it, for example, from infecting a cell), or allows other parts of the immune system to neutralize and destroy the virus.
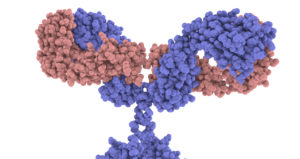
In the diagram above, I drew the antigen as a circle and gave the antigen-binding site on the antibody a complementary shape. But to get a sense of the required specificity of the antigen-binding site, take a look at the image at right, from the Garvan Institute of Medical Research / Centre for Targeted Therapy. This is a space-filling model, with every atom represented (note: I’ve set up the image so you can click on it to open up a new tab with an enlarged version of it: please click it to take a close look at the antigen-binding sites on the tips of the arms). The key point is that this shape, down to the nanometer scale, is specific. It can only do its job (binding with antigen) if it has this exact shape. And this leads to the question: how can protein shape be determined so specifically?
The second feature of protein shape that needs to be explained is that for many proteins, the shape is dynamic. It can change.
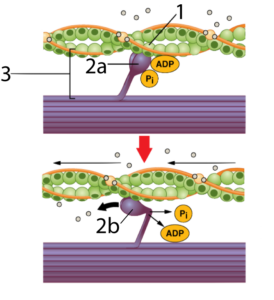 Let’s consider what happens when you contract a muscle. Two interacting proteins are involved: actin (at “1”) and myosin (at “2a” and “2b”). The two interact to form a cross-bridge (at “3”), which is made possible by complementarity in shape between the head of the myosin and a binding site on the actin.
Let’s consider what happens when you contract a muscle. Two interacting proteins are involved: actin (at “1”) and myosin (at “2a” and “2b”). The two interact to form a cross-bridge (at “3”), which is made possible by complementarity in shape between the head of the myosin and a binding site on the actin.
When myosin binds with ATP (the cell’s energy molecule: click here to learn more about how it works), the ATP is broken into its lower energy form (ADP and Pi), which enables the myosin to reach and grab onto actin. When the myosin releases ADP and Pi, it again changes shape, bending into a right angle (as shown at “2b”), which pulls on the actin, shortening the muscle fiber.
In other words, the myosin protein is like a lever: provide it with ATP (and meet a few other conditions) and it will bend. Other protein-based structures spin like propellers.
Take three minutes and watch the video below, created by XVIVO for Harvard University.
Pretty much every mind-blowing thing that’s represented involves the dynamic nature of proteins. So the question is what kind of molecular structure allows a protein (like myosin) to change its shape?
4b. Primary Structure
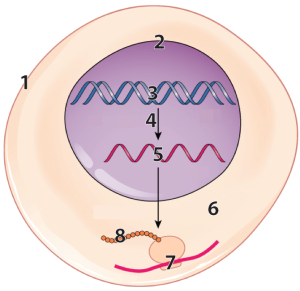
The specificity of protein shape, as well as many proteins’ ability to change shape, emerges from three or four levels of molecular interactions, usually referred to as “levels of structure.” The first level, also called primary structure, is determined genetically.
DNA (“3”) in the nucleus (“2”) of our cells (“1”) has instructions for making RNA (“5”).
That RNA is synthesized through a process called transcription, which is indicated by the arrow at “4.” After transcription, a variety of processes in the nucleus modify the RNA, converting it into messenger RNA (mRNA).
The mRNA leaves the nucleus and enters the cytoplasm (“6”), where ribosomes (“7”) translate the RNA into a polypeptide (“8”).

Note that each of the little circles in “8” above is an amino acid residue. Now, connect that image to what you’ve learned about amino acids, peptide bonds, and polypeptides. “8” above is, biochemically, what you see on the right: a linear sequence of amino acid residues in a polypeptide chain.
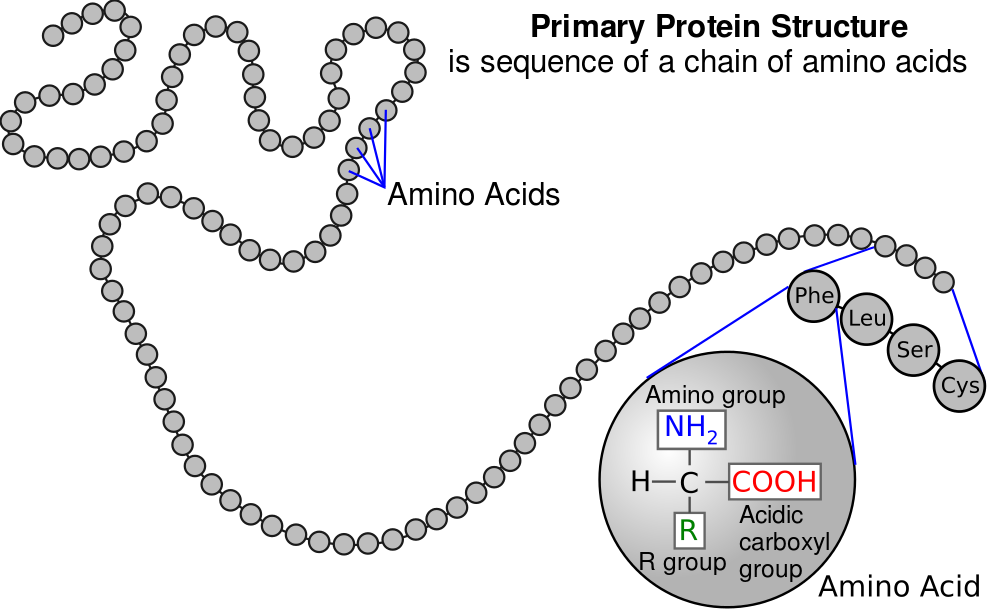 So, now we need to explain how we get from the genetically determined linear string of amino acids in a polypeptide chain to a protein’s three-dimensional shape.
So, now we need to explain how we get from the genetically determined linear string of amino acids in a polypeptide chain to a protein’s three-dimensional shape.
4c. Secondary Structure
The next level of interaction involves hydrogen bonds that form between carbonyl functional groups and amino groups within the backbone of a polypeptide. To see this, take a look at the diagram of a polypeptide below:
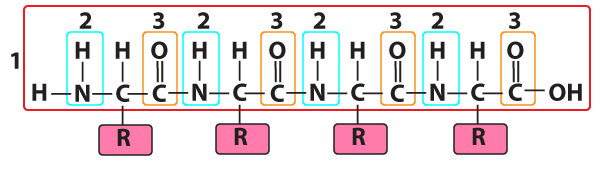
Number “1” is the polypeptide backbone. In the same way that your ribs are attached to your backbone, the side chains/R groups of each amino acid residue are attached to the polypeptide backbone.
The polypeptide was formed by connecting the amino group of one amino acid to the carboxyl group of the next, in a dehydration synthesis reaction that removed an “—H” from the amino group and an “—OH” from the carboxyl group. This leaves behind two recognizable functional groups. At “2” there’s an —NH (still called an “amino group,” despite the loss of a hydrogen atom). At “3” is a carbonyl group. Note that it was a carboxyl group (—COOH), but having lost its terminal —OH, it’s now carbonyl (—C=O).
Both the amino group and the carbonyl group are polar. The amino has a partially positive charge (δ+), and the carbonyl has a partially negative charge (δ–). That means these two functional groups, if they get close enough to one another, can form a hydrogen bond. And while hydrogen bonds are relatively weak (much weaker than the covalent bonds that hold the polypeptide backbone together), they can twist the polypeptide backbone into particular shapes, and stabilize those shapes. The two shapes are called an alpha helix, and a beta-pleated sheet.
4c.1. Alpha Helix
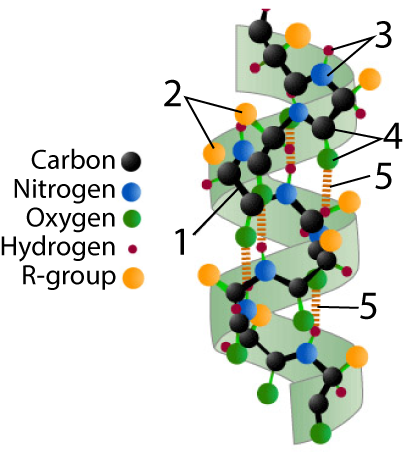
An alpha helix is a corkscrew, stabilized by hydrogen bonds.
On the right, you see one such helix. The polypeptide backbone is indicated by “1.” You should be able to notice the repeating pattern of carbon-carbon-nitrogen within the backbone.
R-groups (shown at “2”) hang off of each central carbon. The nitrogen atoms are bonded to hydrogen atoms, forming amino groups (“3”), while the second carbon atom (the one that’s not the central carbon) is bonded to an oxygen, creating a carbonyl group (“4”).
In an alpha helix, hydrogen bonds (shown at “5” form between the carbonyl group and amino groups in amino acid residues that are four amino acids away from one another.
4c.2. Beta Pleated Sheet
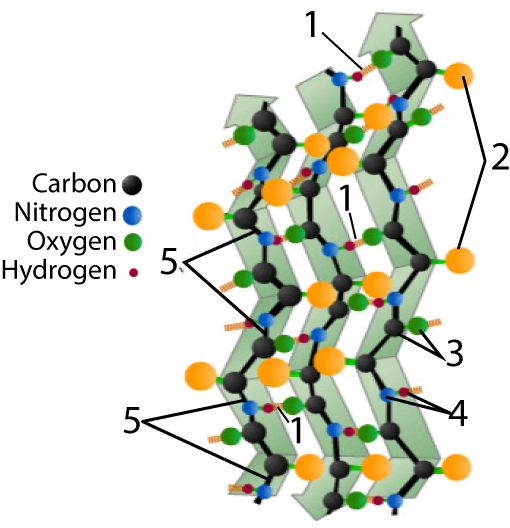 The other secondary structure to know is a beta-pleated sheet.
The other secondary structure to know is a beta-pleated sheet.
4d. Tertiary Structure
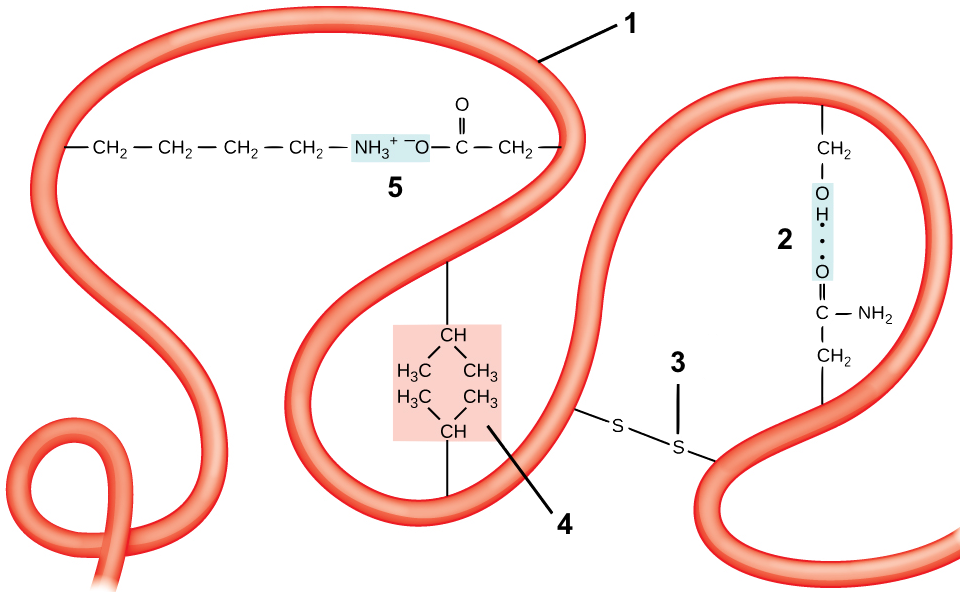
Tertiary structure comes about through interactions between R groups, which cause a polypeptide chain (“1”) to twist and bend. These interactions include
- Hydrogen bonds: if a side chain with a carbonyl group (which has a partially negative charge) gets close to a hydroxyl group or an amino group, a hydrogen bond can form. One such bond between a carbonyl and a hydroxyl is shown at “2.”
- Disulfide Bridges: the amino acid cysteine has a sulfhydryl functional group (—SH) at the end of its side chain. If two of these sulfhydryl groups from cysteines in the same polypeptide chain get close to one another, they can form a disulfide bridge. A disulfide bridge is a covalent bond.
- Hydrophobic interactions: If hydrophobic side chains come into proximity, they can form hydrophobic clusters. These clusters are maintained by two forces. The first is the exclusion of these hydrophobic side chains from the hydrogen bonding between water molecules in the surrounding solution. The second is the weak Van Der Waals bonds that form between these hydrophobic side chains. One such hydrophobic cluster is shown at “4.”
- Ionic bonds: These form when side chains with full positive charges come into proximity with side chains that have full negative charges, as is shown in “5.”
4e. Quaternary Structure
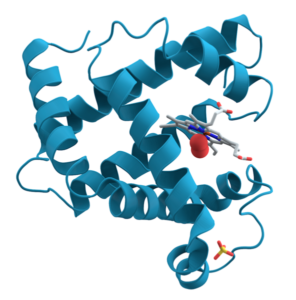
For some proteins, primary, secondary, and tertiary interactions explain the protein’s form and function. Myoglobin, for example, is a protein that stores and releases oxygen within muscle tissue. It consists of 154 amino acids that twist, turn, and coil into the shape shown at the right. As you look, I’m hoping that you’re noting the alpha helices (and connecting them to secondary structure) and hairpin turns (and thinking that some tertiary bond must be causing the polypeptide chain to turn at such a sharp angle).
In other proteins, there’s a fourth level of structure, called quaternary. Quaternary structure involves two or more folded polypeptide chains interacting to form a more complex structure.
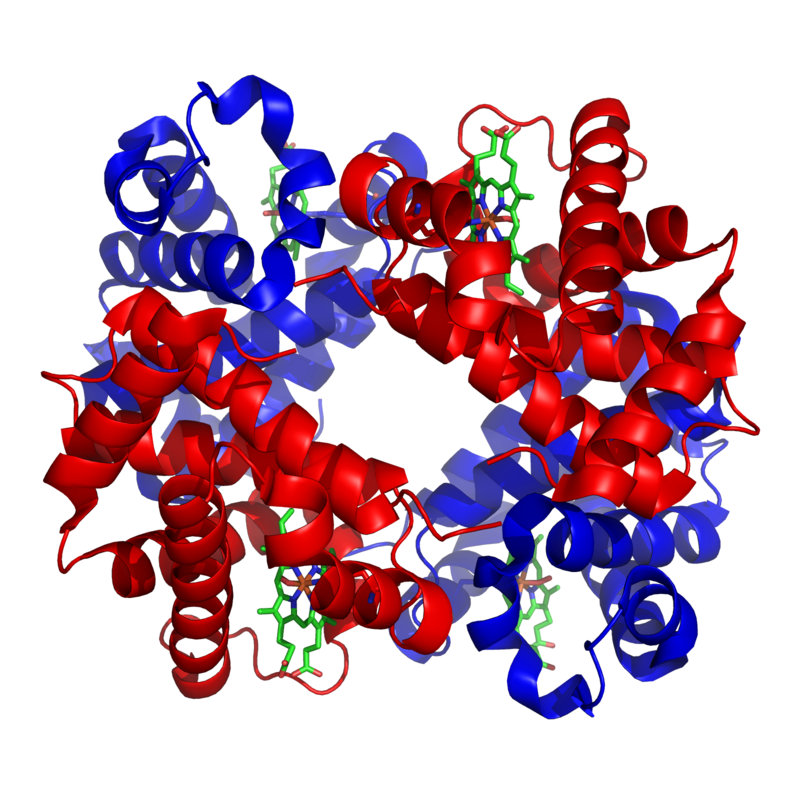
An example of one such quaternary-level protein is hemoglobin. Hemoglobin is the molecule that carries oxygen in our red blood cells. It consists of four polypeptide chains, two alpha chains (shown in red), and two beta chains (shown in blue).
The bonds between the polypeptide subunits in a quaternary protein can include any of the bonds that appear at the tertiary level: hydrogen bonds, hydrophobic interactions, disulfide bridges, and ionic bonds.
4f. The Dynamic Nature of Proteins
My goal in the sections above was to explain how proteins have such specific shapes. But the second question we wanted to answer about proteins was how they can change their form to act as molecular versions of gates, levers, pulleys, vice grips, propellers, and so on.
 The basic idea is that when proteins bind with other molecules, when their environment changes (through temperature changes or pH changes), or when they are compressed or stretched, the configuration of bonds within that protein can change. Changing the configuration of bonds, in turn, can change the protein’s shape.
The basic idea is that when proteins bind with other molecules, when their environment changes (through temperature changes or pH changes), or when they are compressed or stretched, the configuration of bonds within that protein can change. Changing the configuration of bonds, in turn, can change the protein’s shape.
An alpha helix, for example, is like a molecular spring. A change in another part of the protein can compress that spring, or stretch it out, after which it might return to its original form. Or, if a tertiary bond that caused a hairpin turn in a polypeptide chain is altered, then that chain might straighten out, only to snap back later.
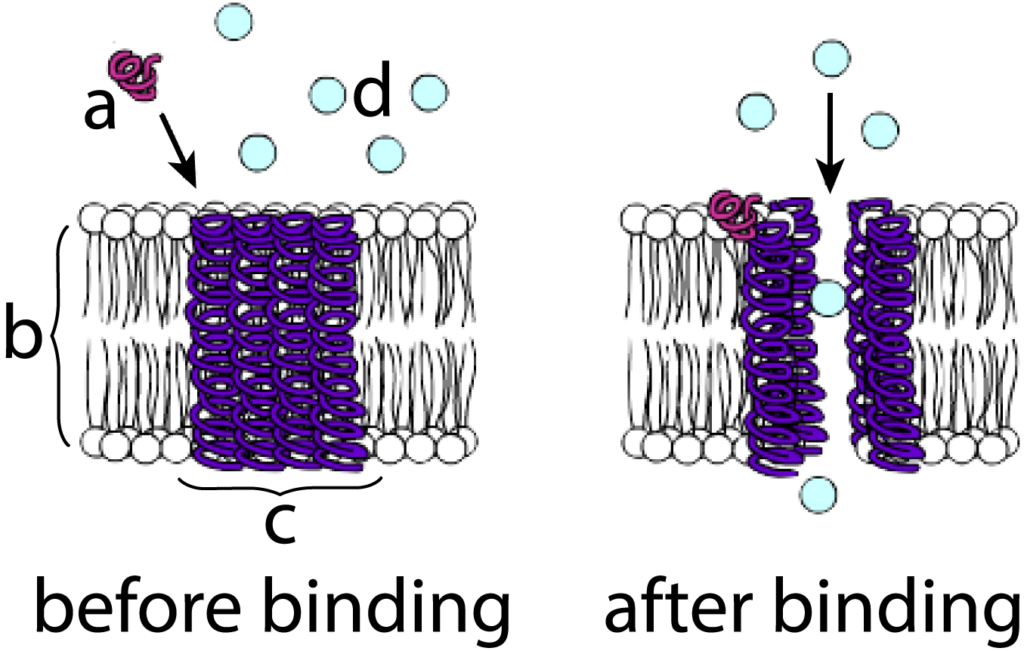
Let’s use the example of a membrane protein that acts as a calcium gate as an example. The diagram at left shows a protein hormone (“a”) diffusing toward the cell membrane (“b”). Embedded within the membrane is a channel protein (“c”). In such membrane channels, alpha helices are often a predominant structure, and you should be able to see several of these helices spanning the membrane.
The binding of the hormone with the channel protein (shown at right), changes the configuration of these alpha-helices so that they move apart from one another. This creates an opening in the membrane where one didn’t exist before. Substances can now diffuse through this opening. When the hormone, which is only loosely bound to the channel protein through hydrogen bonds, dissociates from the channel, the channel will close up, and diffusion will stop.
5. Denaturation
A protein’s function is based on its structure. As we’ve seen above, the interactions that result in protein structure at the secondary, tertiary, and quaternary levels involve some forces that are relatively weak, such as hydrogen bonds and other Van Der Waals bonds. Other interactions, such as ionic bonds, only occur if a solution has a specific pH. Proteins, in a word, can be delicate, and their function can be affected by changes in their environment. A protein that loses its function because of environmental change —usually heat or pH — is said to be denatured, and the process by which this happens is called denaturation. A quick look at enzymes will show you how this works (you can learn more about enzymes here).
Enzymes are catalysts that speed up chemical reactions in living things. With very few exceptions, enzymes are proteins. Here’s an example of how an enzyme might work to break the disaccharide lactose into two monosaccharides, glucose and galactose
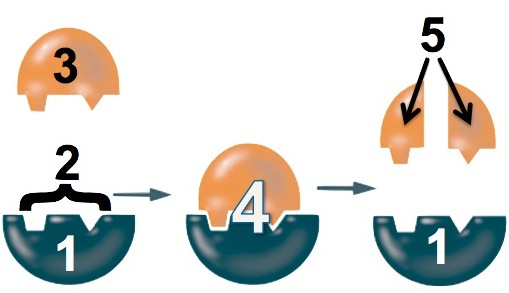
- The enzyme (“1”) binds with the lactose (“3”). The binding is only possible because a specific part of the enzyme, its active site (“2”), has a shape that complements the shape of the lactose.
- While the enzyme is bound to the lactose, the enzyme changes shape to stress the bonds that hold the glucose and galactose monomers together.
- The bond breaks and glucose and galactose are released from the enzyme.
While the details of different enzyme-catalyzed reactions will vary, the key point is that the enzyme can catalyze the reaction only if it can fit together with its substrate (whatever molecule it interacts with). That fit is as specific as the ability of a key to open a lock. Change the key’s shape, and the lock won’t open.
With these ideas in hand, let’s look at the graph below, which shows the relationship between the rate of an enzyme-catalyzed reaction, the percentage of active enzyme molecules, and temperature.
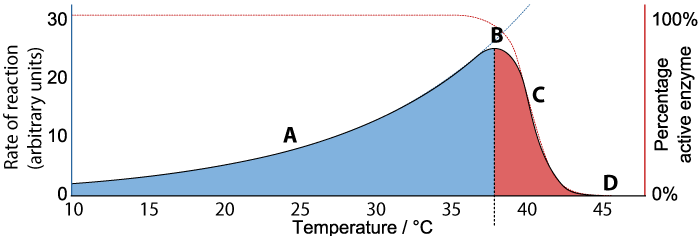
We’ll consider the rate first. As the temperature increases from 10° C to about 37°C (“A”), the rate of the reaction also increases. At about 37°C, the reaction reaches a peak (“B”). As the temperature continues to increase much beyond 37°C, the rate of the reaction declines (“C”) until at 45°C the rate of the reaction falls to zero.
Now consider the “percentage of active enzyme” (the Y-axis on the right). For any temperature between 10° C to just beyond 37°C, the percentage of active enzyme is 100%. But after point “B,” the percentage drops off (“C”) until it reaches zero (“D”).
As I’m sure you’ve surmised, what’s happening is denaturation. Up to a certain temperature, the enzyme can hold its shape and interact with its substrate to bring about the reaction. As temperature increases up to 37°C, the reaction proceeds faster and faster, because higher temperature means more molecular movement and a greater chance that the enzyme will bump into its substrate at the right orientation to bring the reaction about. Beyond 37°C, the kinetic energy in the system starts to disrupt the bonds that are holding the enzyme in its required three-dimensional shape. As this shape starts to change, the enzyme becomes unable to bond with its substrate, and the rate of the reaction (along with the amount of active enzyme) falls to zero.
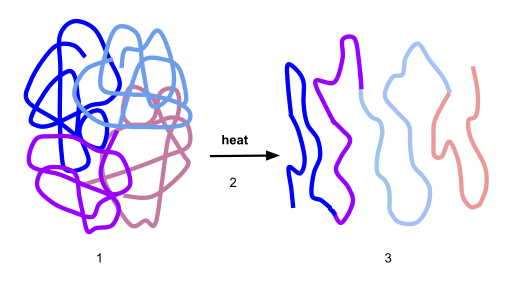 Here’s a visual representation of denaturation. Number “1” shows the protein in its optimal conformation. Heat (“2”) breaks the bonds that stabilize this protein’s shape at the secondary and tertiary levels. As a result, the enzyme unfurls into a polypeptide.
Here’s a visual representation of denaturation. Number “1” shows the protein in its optimal conformation. Heat (“2”) breaks the bonds that stabilize this protein’s shape at the secondary and tertiary levels. As a result, the enzyme unfurls into a polypeptide.
Sometimes, like when you cook an egg and change liquid albumin into solid egg white, the denaturation is irreversible. In other words, you might let your egg white cool down, but it’s not going to liquefy again. On the other hand, some proteins can temporarily be denatured (usually by changes in pH). When the pH is reset to the original level, the amino acids in the polypeptide might be able to interact in a way that restores the protein’s original shape. That process is called renaturation.
6. Proteins Quiz
[qwiz use_dataset=”SMV_biochem_8_proteins_cumulative” dataset_intro=”false” qrecord_id=”sciencemusicvideosMeister1961-SMV-Proteins”] [h]
Proteins Quiz 2
[i]
[x][restart][/qwiz]
Links
- Nucleic Acids Overview (the next tutorial in this series)
- Biochemistry final flashcards and quiz
- Biochemistry Main Menu
