1. After Making Monomers, Things Gets Harder
We saw in the last tutorial how, based on the work of Stanley Miller and many others, it seems that monomers could have formed abiotically on Earth or in space. But the next steps are harder, and few (if any) have been definitively recreated in laboratory simulations. To review these steps, fill in the blanks in the table below.
[qwiz style=”width:650px” qrecord_id=”sciencemusicvideosMeister1961-Steps in the Origin of Life, Table 2″]
[h]Steps in the Origin of Life, Table 3 (with follow-up questions)
[i]Another Origin of Life Haiku
Primordial Soup
A warm stew of monomers
Could this be the start?
[q]
| FIRST | [hangman] creation of monomers (the molecular building blocks of life) | |||
| NEXT | Abiotically link monomers to form [hangman] | Origin of [hangman]. Create a system for processing matter and energy and for removing wastes. | Origin of [hangman]. Provide the system with a way to pass on instructions for maintenance, growth, and reproduction | Encapsulate the system with a [hangman] to keep it from dissolving away, creating the first primitive [hangman]. |
[c]YWJpb3RpYw==[Qq]
[c]cG9seW1lcnM=[Qq]
[c]bWV0YWJvbGlzbQ==[Qq]
[c]aGVyZWRpdHk=[Qq]
[c]bWVtYnJhbmU=[Qq]
[c]Y2VsbHM=[Qq]
[q multiple_choice=”true”] Based on what you know about biology, the first self-replicating informational molecule would probably be a(n) __________ acid.
[c]IGFtaW5v[Qq]
[f]IE5vLiBBbWlubyBhY2lkcyBhcmUgdGhlIG1vbm9tZXJzIG9mIHByb3RlaW5zLiBUaGV5JiM4MjE3O3JlIG5vdCBpbmZvcm1hdGlvbmFsLg==[Qq]
[c]IG51Y2 xlaWM=[Qq]
[f]IEV4Y2VsbGVudC4gSW4gbGlmZSB0b2RheSwgaW5mb3JtYXRpb25hbCBtb2xlY3VsZXMgYXJlIG51Y2xlaWMgYWNpZHM6IGVpdGhlciBETkEgb3IgUk5BLg==[Qq]
[c]IGZhdHR5[Qq]
[f]IE5vLiBGYXR0eSBhY2lkcyBhcmUgY29tcG9uZW50cyBvZiBsaXBpZHMuIFRoZXkmIzgyMTc7cmUgbm90IHVzZWQgZm9yIGluZm9ybWF0aW9uLCBhbmQgaXQmIzgyMTc7cyBoYXJkIHRvIHNlZSBob3cgdGhleSBjb3VsZCBiZS4=[Qq]
[q multiple_choice=”true”] From the choices below, the type of molecule that would make up an encapsulating membrane would probably be primarily composed of _________ acids.
[c]IGFtaW5v[Qq]
[f]IE5vLiBBbWlubyBhY2lkcyBhcmUgdGhlIG1vbm9tZXJzIG9mIHByb3RlaW5zLiBUaGV5IGFyZSBjcnVjaWFsbHkgaW1wb3J0YW50IHBhcnRzIG9mIG1lbWJyYW5lcywgYnV0IHRoZXkmIzgyMTc7cmUgbm90IHRoZSBtYWluIHN0cnVjdHVyYWwgY29tcG9uZW50IG9mIG1lbWJyYW5lcy4=[Qq]
[c]IG51Y2xlaWM=[Qq]
[f]IE5vLiBJbiBsaWZlIHRvZGF5LCBpbmZvcm1hdGlvbmFsIG1vbGVjdWxlcyBhcmUgbnVjbGVpYyBhY2lkcy4gWW91JiM4MjE3O3JlIGxvb2tpbmcgZm9yIGEgbW9sZWN1bGUgdGhhdCB3b3VsZCBtYWtlIHVwIG9uZSBvZiB0aGUgY29tcG9uZW50cyBvZiBtZW1icmFuZXMu[Qq]
[c]IGZh dHR5[Qq]
[f]IEV4Y2VsbGVudC4gRmF0dHkgYWNpZHMgYXJlIGEga2V5IGNvbXBvbmVudCBvZiB0aGUgbW9sZWN1bGVzIHRoYXQgbWFrZSB1cCBtZW1icmFuZXMgKHBob3NwaG9saXBpZHMgaW4gYmFjdGVyaWEgYW5kIGV1a2FyeWEsIGEgcmVsYXRlZCBsaXBpZCBtb2xlY3VsZSBpbiBhcmNoYWVhKS4=[Qq]
[/qwiz]
Moving beyond the abiotic creation of monomers, the science becomes more speculative. Here are just two of the problems we’ll have to resolve.
- How were the first polymers created on an enzyme-free, watery Earth? Life is based on molecules with complex shapes that can catalyze the anabolic (building up) and catabolic (breaking down) reactions that happen in cells. These catalytic molecules are polymers and include enzymes (which are polymers of amino acids) and RNAs (which are polymers of ribonucleotides). Polymers are also how life stores hereditary information, a role carried out mostly by the nucleic acid polymer DNA, though some viruses use RNA.
Here’s the problem: to make proteins and RNA, life needs proteins and RNA. To make proteins, for example, cells use ribosomes, a two-unit structure that consists of thousands of RNA nucleotides (and dozens of amino acids) organized into a finely tuned molecular machine. To synthesize RNA, cells use the enzyme RNA polymerase, a protein consisting of hundreds of amino acids. So, how, during the emergence of life, could RNA and proteins form in the absence of already existing RNA and proteins?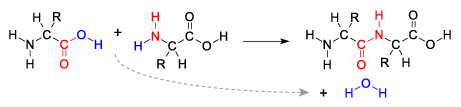
A dehydration synthesis reaction An additional problem related to polymer creation has to do with the relationship between the process of polymer formation and water. It’s a bit of a paradox because life is dependent on water, but the complex molecules of life don’t easily form in water — at least, not without a lot of enzymatic help. Monomers form into polymers through dehydration synthesis reactions. In these reactions, water molecules are formed as a hydrogen atom from one monomer and a hydroxyl group from another are pulled out by an enzyme, which simultaneously catalyzes the bond between the two monomers. Polymers break apart into monomers through the opposite process, hydrolysis, during which an enzyme splits a water molecule apart, adding hydrogen to one of the resulting monomers, and a hydroxyl group to the other one. The key point is that the presence of water works against dehydration synthesis, and promotes hydrolysis. Since the surface of the early Earth was a very wet place, it’s hard to see how, in the absence of enzymes, polymers could spontaneously form and accumulate.
-
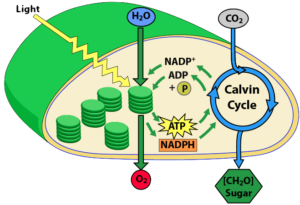
A chloroplast, showing the reactions of photosynthesis. Click to enlarge. In the absence of photosynthesis, what energy source could have been channeled to create the first living things? Life is highly ordered, and creating order requires energy. With one important exception (hydrothermal vent systems, which we’ll explore below), almost all life today is powered by sunlight. Unlike other forms of electromagnetic radiation, sunlight has enough energy to drive the reactions of photosynthesis, but not so much energy that it would destroy molecules like protein or DNA (which is what ultraviolet or X-ray radiation does). As life was evolving, what consistent, powerful, non-destructive energy source could have played sunlight’s role?
As we look at where and how life could have emerged, we’ll have to be able to resolve these (and other) questions in a satisfactory way.
2. Darwin’s “Warm Little Pond,” and a few other Speculations about Life’s Cradle
2a.Problems with the Pond
In 1871, Charles Darwin wrote to his friend, the botanist Joseph Hooker (Darwin Correspondence).
But if (& oh what a big if) we could conceive in some warm little pond with all sorts of ammonia & phosphoric salts,—light, heat, electricity [and so on being] present, that a protein compound was chemically formed, ready to undergo still more complex changes…
This was Darwin’s version of the primordial soup. It set the stage for the Oparin-Haldane hypothesis, and Miller-Urey’s attempt to test that hypothesis. But if an essential stage in the evolution of life is the formation of polymers from monomers, there are a couple of reasons why warm ponds, tidepools, or shallow seas were unlikely places for that to have happened.
- The was very little land on which ponds or tide pools could have formed. The early Earth was covered by one global ocean, punctuated by a few volcanic islands.
- From 4.5 to about 3.8 bya, the Earth was in a period of heavy bombardment. Earth’s surface was being pummeled by comets and meteors, sterilizing the impact zones.
- The much closer Moon would have created enormous tides that would have washed any emerging concentration of monomers in a warm little pool out to sea. As a result, pools of concentrated monomers were unlikely, and without concentrated monomers, it’s hard to imagine monomers linking together to form polymers.
- If that weren’t enough, re-read the material above about dehydration synthesis and polymer formation. Put monomers together in water, and the last thing they’ll spontaneously do is combine.
- There’s no energy source. As biochemist Nick Lane points out in Life Ascending, The Ten Great Inventions of Evolution, the primordial soup is “thermodynamically flat.” Randomly run energy through such a soup and the most likely thing to happen is that the molecules inside will break down, not grow in complexity.
2b. Directed Panspermia
These difficulties have led to some alternative speculations about where life might have begun. Francis Crick, the co-discoverer of DNA, and Leslie Orgel, an eminent origin of life experimenter and an originator of the RNA world hypothesis (discussed in the next tutorial), speculated that life might have arrived on Earth through directed panspermia. The idea is that an alien civilization wanted to seed the universe (or at least our galaxy) with life. To do so, they filled rockets with bacteria and sent them into space. One of these rockets landed on Earth billions of years ago, seeding the Earth with life. If that leaves you wondering how life evolved on the alien’s planet, it’s a good question. You can read Crick and Orgel’s speculations here.
2c.The Case for a Martian Cradle

Another idea is that life originated on Mars. Mars had shallow seas early in its history. But Mars has always been a drier world than Earth. In this scenario, abiotically formed monomers in drying pools on Mars could have formed into polymers through dehydration synthesis, a process that would have been coaxed along by inorganic mineral catalysts. A series of linked pools on Mars would have acted as reaction chambers, with polymers becoming increasingly concentrated, interacting with one another, and eventually becoming complex enough to become self-replicating molecules. These would have become encapsulated in cells, which would develop a metabolism, and evolve into the first bacteria.
How would these bacteria get to Earth? Over 100 meteorites on Earth have been identified as being of Martian origin. This happens when a meteor strikes Mars and blasts Martian rocks into space. These Martian rocks then become meteors, which spiral in toward Earth. If one of these Martian rocks harbored bacteria, and if these bacteria survived the trip through space and their crash-landing on Earth, then Martian life could have taken hold here. That means that both you and I (according to this scenario) would have Martian ancestors. (See this article in Smithsonian Magazine, this much more detailed journal article Mars, Panspermia, and the Origin of Life: Where Did it All Begin, or read pages 58 to 60 in A New History of Life, Peter Ward and Joe Hirschvink).
As we’ll see below, there are some viable and reasonable Earthly locations and scenarios for the origin of life. But first, take the following quiz.
3. Difficulties in Polymer Formation: Checking Understanding
[qwiz qrecord_id=”sciencemusicvideosMeister1961-Difficulties with Polymer Formation”]
[h]Difficulties in Polymer Formation; Warm Little Ponds and other Speculations
[i]Another Origin of life Haiku
A Warm Little Pond,
Pummeled by Meteor Strikes.
Not a nice cradle.
[q] A problem associated with any account of abiotic polymer formation is that in living systems today, monomers form into polymers through[hangman] synthesis reactions, which are catalyzed by [hangman]
[c]IGRlaHlkcmF0aW9u[Qq]
[c]IGVuenltZXM=[Qq]
[q] In today’s living systems, amino acids are polymerized through the action of a two-unit molecular machine called a [hangman]. Any origin of life scenario needs to be able to explain how with these being absent, [hangman], the polymers of amino acids, could still form.
[c]IHJpYm9zb21l[Qq]
[c]IHByb3RlaW5z[Qq]
[q] Since life’s emergence, almost all life has been powered by [hangman], which has provided the energy for chloroplasts (and certain bacteria) to carry out [hangman].
[c]IHN1bmxpZ2h0[Qq]
[c]IHBob3Rvc3ludGhlc2lz[Qq]
[q] Visible light is powerful enough to power photosynthesis. Unlike lightning, visible light doesn’t destroy molecules like [hangman] or protein.
[c]IEROQQ==[Qq]
[q multiple_choice=”true”] According to Darwin, life possibly started
[c]IGluIHRoZSBoaWdobHkgcmVkdWNlZCBhdG1vc3BoZXJlIG9mIEVhcmx5IHBsYW5ldCBlYXJ0aC4=[Qq]
[f]IE5vLiBUaGF0IHdhcyBtb3JlIGxpa2UgU3RhbmxleSBNaWxsZXImIzgyMTc7cyBpZGVhLg==[Qq]
[c]IGluIGEgd2FybSBs aXR0bGUgcG9uZC4=[Qq]
[f]IEV4Y2VsbGVudC4gVGhhdCB3YXMgRGFyd2luJiM4MjE3O3MgaWRlYSBhYm91dCB0aGUgb3JpZ2luIG9mIGxpZmUu[Qq]
[c]IEluIHRoZSBib2lsaW5nIEhvdCBTcHJpbmdzIGluIHBsYWNlcyBsaWtlIEdyZWVubGFuZCwgb3IgWWVsbG93c3RvbmUgTmF0aW9uYWwgUGFyay4=[Qq]
[f]IE5vLiBJdCYjODIxNztzIG5vdCBsaWtlbHkgdGhhdCBEYXJ3aW4sIHdpdGggdGhlIHRvb2xzIGhlIGhhZCBhdmFpbGFibGUgYXQgaGlzIHRpbWUsIGNvdWxkIGV2ZW4gY29uY2VpdmUgb2YgbGlmZSBleGlzdGluZyBpbiBob3Qgc3ByaW5ncy4=[Qq]
[q multiple_choice=”true”] Which of the following is the least probable reason why life is unlikely to have first emerged in ponds or tidepools at or near the surface of the Earth?
[c]IExhcmdlIGNvbWV0cyBhbmQgbWV0ZW9ycyB3ZXJlIHN0cmlraW5nIHRoZSBFYXJseSBlYXJ0aCwgbWFraW5nIGxpZmUgaW1wb3NzaWJsZS4=[Qq]
[f]IE5vLiBUaGF0IFdBUyBoYXBwZW5pbmcgdG8gdGhlIHN1cmZhY2Ugb2YgdGhlIGVhcmx5IGVhcnRoLiBGaW5kIHRoZSBsZWFzdCBwcm9iYWJsZSByZWFzb24u[Qq]
[c]IFRoZXJlIHdhcyB2ZXJ5IGxpdHRsZSBsYW5kLCBtZWFuaW5nIHZlcnkgZmV3IHRpZGUgcG9vbHMgb3IgcG9uZHMgd2hlcmUgbGlmZSBjb3VsZCBoYXZlIHN0YXJ0ZWQu[Qq]
[f]IE5vLiBUaGF0IGlzIHRydWUgb2YgdGhlIGVhcmx5IGVhcnRoLiBGaW5kIHNvbWV0aGluZyB0aGF0IHdhc24mIzgyMTc7dCB0cnVlLg==[Qq]
[c]IFRoZSB5b3VuZyBzdW4gaGFkIHRvbyBtdWNoIG VuZXJneSB0byBhbGxvdyBsaWZlIHRvIGZvcm0=[Qq]
[f]IENvcnJlY3QuIFRoZSBlYXJseSBzdW4gd2FzIHdlYWtlciB0aGFuIHRvZGF5JiM4MjE3O3Mgc3VuLCBhbmQgbm90aGluZyBhYm91dCB0aGF0IHN1biB3b3VsZCBoYXZlIHByZXZlbnRlZCB0aGUgZm9ybWF0aW9uIG9mIGxpZmUgaW4gdGlkZXBvb2xzIChidXQgbWFueSBvdGhlciB0aGluZ3Mgd291bGQgaGF2ZSku[Qq]
[q] The difficulty of envisioning a scenario for life’s emergence on Earth led Francis Crick (co-discoverer of the structure of [hangman] and Leslie Orgel to hypothesize that life was inoculated on earth through alien rockets, an idea known as directed [hangman].
[c]IEROQQ==[Qq]
[c]IHBhbnNwZXJtaWE=[Qq]
[q] Because of the difficulties of polymer formation on the watery early Earth, some scientists have theorized that life might have begun on [hangman] and then subsequently transported to earth on [hangman].
[c]IE1hcnM=[Qq]
[c]IG1ldGVvcnM=[Qq]
[/qwiz]
4. Alkaline Hydrothermal Vents are a Possible Site for Life’s Origins
4a. Black Smoker Vents Support Diverse Ecosystems

In 1977, deep-sea hydrothermal vent ecosystems were discovered. These ecosystems are not powered by sunlight. Found at depths of 2500 meters below the surface of the ocean (and much deeper), they are powered by molten magma rising up toward the ocean floor. In these areas, seawater percolates into the ocean floor, becomes superheated, and then rises full of dissolved minerals like iron sulfides. Deposition of these and other minerals leads to the formation of chimneys that emit a continuous stream of black, smokey water (see the photo at left). As a result, these vents are called “Black Smokers.”
These Black Smoker vents support biological communities with population densities that rival those in coral reefs or tropical rainforests. At the base of these food chains are bacteria that can perform chemosynthesis. Chemosynthesis parallels photosynthesis, with the important difference of using hydrogen sulfide (H2S) in place of water (H2O). Here’s the chemosynthetic reaction that occurs at the vents.
12H2S + 6CO2 → C6H12O6 (carbohydrate) + 6H2O + 12S
The carbohydrate formed sustains giant tube worms (which harbor these chemosynthetic bacteria in an organ inside their bodies, and have lost their mouths, gut, and anus). Shrimp graze on mats of these bacteria. The vents also support clams, mussels, and crabs, each of which has some type of relationship with the chemosynthetic bacteria. If you want to explore the biology of these Black Smoker Vents in more detail, click the following link for an amazing video with science journalist Ed Yong about the biology of vent tube worms.
Upon their discovery, origin of life researchers began speculating that these hydrothermal vents, with their constant flow of energy and their sheltered location — far away from the tides and meteor strikes that were occurring at the early Earth’s surface — could have been life’s cradle. But a second type of undersea vent, discovered in the year 2000, has properties that would have been even more favorable for serving as a site for the origin of life. These are called alkaline hydrothermal vents, and here’s how they work.
4b. Alkaline Hydrothermal Vents
Alkaline Hydrothermal vents are also powered by geothermal energy, but less directly than the Black Smoker vents described above. The first of these vents to be discovered, called the Lost City, is located about 20 kilometers away from the mid-Atlantic ridge. It was discovered by a team from the University of Washington, led by Deborah Kelley.

In the diagram at left, you can see the overall geography of these vents. Number 1 shows rising magma. The magma’s rise causes seafloor spreading and creates structures like the mid-Atlantic Ridge and its rift valley (shown at “3”). Number “2” is a black smoker vent. At “4” is an alkaline hydrothermal vent.

Like black smoker vents, the Lost City vents are created by mineral deposition. Chimneys composed of calcium carbonate grow up to 60 meters tall. Water leaving the vents is hot (40° C to 90° C), but nothing like the 400° C fluid emerging from black smoker vents. As their name indicates, the fluids emerging from these vents are alkaline (pH 9 to 11, as opposed to the slightly acidic effluent from Black Smoker Vents). They’re also rich in highly reduced compounds, including molecular hydrogen (H2) and methane (CH4). The Lost City spires are also much longer lived than Black Smokers: radiometric dating sets the age of the Lost City system as at least 30,000 years old, making these vents hundreds of times older than Black Smoker Chimneys.
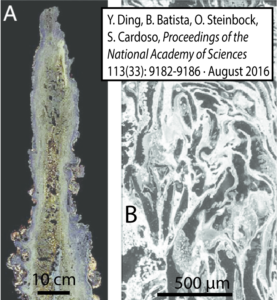
Another important difference between alkaline hydrothermal vents and Black Smoker vents is their internal structure. Alkaline hydrothermal vents are riddled with tiny interconnected pores, some of which are as small as a micrometer. Note that this scale is within the range of the size of bacterial cells, which range from 0.5 to 5 micrometers in diameter.
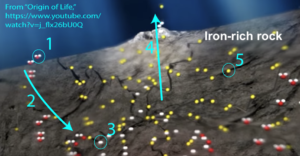
Driving the chemistry at the vents is a reaction called serpentinization. The fracturing rock, composed of a mineral called olivine (magnesium iron silicate), chemically reacts with seawater (“1”). The reaction is exergonic and results in the formation of a new mineral called serpentine, along with the release of hydrogen gas (hydrogen is at “3”), alkaline fluids, and heat.
4c. Why Alkaline Hydrothermal Vents May Have Been Life’s Hatchery
In ancient Earth, there would be two additional features of the vents that aren’t seen today. First, today’s seas are alkaline: the average pH of seawater is about 8.2. In Archaean times, before the advent of photosynthesis (which absorbs carbon dioxide), the oceans would have been acidic. To review, acidic means more hydrogen ions (H+) than hydroxide ions (OH–); alkaline or basic means the reverse). Second, the ancient seas had large amounts of dissolved iron. Put all of this together, and according to biologists like Mike Russell (from NASA’s Jet Propulsion Laboratories), William Martin (University of Dusseldorf), and Nick Lane (University College, London), the stage would have been set for the alkaline vent environment to have served as the perfect incubator for the emergence of life.
Let’s list the components of these vents, as they would have existed on early Earth.
1. At thousands of meters below the surface of the ocean, they provided a sheltered environment, safe from the heavy bombardment the Earth was experiencing in Archaean times.
2. They provided a consistent flow of gentle energy (which parallels the flow of sunlight that powers life today).
3. They provided a source of highly reduced electrons. Hydrogen gas is rare on Earth, yet it’s constantly released from alkaline vents. This amounts to early life gaining the product of photosynthesis (highly reduced compounds), without all of the highly structured membrane-bound machinery involved in photosynthesis. At the vents, the hydrogen (along with its chemical energy and reducing power) is provided for free.
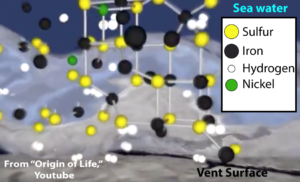
4. The dissolved iron in seawater, along with other metals like nickel, would have combined with sulfur, creating iron-sulfur clusters. These clusters have catalytic properties, and often form the catalytic core of key enzymes that are still at work in living cells today.
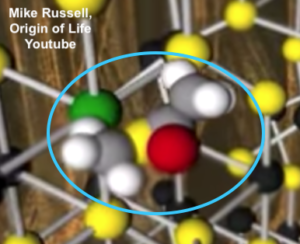
5. These catalytic metals could have promoted the combination of the hydrogen gas emanating from the vents with carbon dioxide dissolved in the seawater, creating reduced carbon compounds like methane (CH4) or acetate (C2H3O2–). Further catalytic reactions could promote the formation of more complex molecules.
The molecule shown to the right is an acetyl thioester, formed from a combination of methane, sulfur, carbon monoxide, and another methane. One acetyl thioester that you’ve probably learned about when studying respiration is acetyl-CoA, which is the highly reduced molecule that diffuses from the cytoplasm to the mitochondria, powering the Krebs cycle. At least one eminent origin of life researcher, Nobel Laureate Christian de Duve, has built an entire theory about the origin of life around the idea of a thioester world, where thioesters played the role that ATP plays in modern life.

6. The porous structure of the alkaline vents would create microscopic chambers that could concentrate these monomers, promoting polymer formation. The formation of polymers would be further promoted by catalytic minerals lining the pores within the vents.
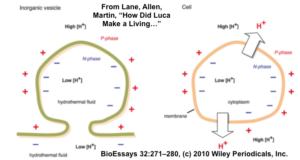
7. The difference in pH between the alkaline fluids emerging from the vents and early Earth’s acidic seawater would have created a proton (H+) gradient. There would have been a higher concentration of protons outside the vents (in the acidic seawater), and a lower concentration of protons in the alkaline vent fluid. Protons would be pulled across breaks in the walls of the vent microchambers, creating proton-motive force. This is another flow of energy, like water flowing down a turbine, ready to be harnessed by emerging life. In all cells today, this flow powers the synthesis of ATP as protons diffuse through the ATP synthase channel.
The image above and to the right, from “How did LUCA Make a Living? Chemiosmosis in the Origin at Life,” by Lane, Allen, and Martin, shows the parallel between proton gradients in hypothetical vesicles in Hadean/Archaean alkaline hydrothermal vents, and those in modern cells.
5. Origin of Life Video: Mike Russell and Bill Martin
Watch the video below, which pulls all of this together. The video stops at 3:07. In the next tutorial, we’ll return to these vents to dig deeper into issues relating to the origins of heredity (the “RNA World”) and the origins of cell membranes.
6. Alkaline Hydrothermal Vents: Checking Understanding
[qwiz qrecord_id=”sciencemusicvideosMeister1961-Alkaline Hydrothermal Vents, Quiz 1″]
[h]Alkaline Hydrothermal Vents
[i]
[q] Both “black smoker” vents and alkaline hydrothermal vents are powered by molten [hangman] rising toward the ocean floor.
[c]IG1hZ21h[Qq]
[f]IEdvb2Qh[Qq]
[q multiple_choice=”true”] Of the two hydrothermal vents discussed above, the type with hotter temperatures are
[c]IGFsa2FsaW5lIGh5ZHJvdGhlcm1hbCB2ZW50cw==[Qq]
[f]IE5vLiBUaGUgYWxrYWxpbmUgdmVudHMgYXJlIHNpZ25pZmljYW50bHkgY29vbGVyIHRoYW4gQmxhY2sgU21va2VyIHR5cGUgdmVudHM=[Qq]
[c]IEJsYWNrIHNtb2 tlciB2ZW50cw==[Qq]
[f]IEV4Y2VsbGVudC4gVGhlIGVmZmx1ZW50IGF0IGJsYWNrIHNtb2tlciB2ZW50cyBjYW4gYmUgdXAgdG8gNDAwIEM=[Qq]
[q] Black smoker-type vents support a food chain that is not based on photosynthesis, but on [hangman].
[c]IGNoZW1vc3ludGhlc2lz[Qq]
[f]IEdvb2Qh[Qq]
[q] At alkaline hydrothermal vents, a reaction with rock called serpentinization causes the release of [hangman] gas into the seawater surrounding the vents.
[c]IGh5ZHJvZ2Vu[Qq]
[f]IENvcnJlY3Qh[Qq]
[q] In the diagram below, an alkaline hydrothermal vent is indicated by number
[textentry single_char=”true”]
[c]ID Q=[Qq]
[f]IENvcnJlY3QhwqA=TnVtYmVyIDQgc2hvd3MgYW4gYWxrYWxpbmUgdmVudC4=[Qq]
[c]IEVudGVyIHdvcmQ=[Qq]
[f]IE5vLCB0aGF0JiM4MjE3O3Mgbm90IGNvcnJlY3Qu[Qq]
[c]ICo=[Qq]
[f]IE5vLiBOb3RlIHRoYXQgdGhlc2UgYWxrYWxpbmUgaHlkcm90aGVybWFsIHZlbnRzIGFyZSBhbHNvIGtub3duIGFzICYjODIyMDtvZmYgcmlkZ2UmIzgyMjE7IHZlbnRzLg==[Qq]
[q] In the diagram below, a black smoker-type vent is indicated by number
[textentry single_char=”true”]
[c]ID I=[Qq]
[f]IEdvb2Qh[Qq]
[c]IEVudGVyIHdvcmQ=[Qq]
[f]IE5vLCB0aGF0JiM4MjE3O3Mgbm90IGNvcnJlY3Qu[Qq]
[c]ICo=[Qq]
[f]IE5vLiBUaGUgYmxhY2sgc21va2VyIHZlbnRzIGFyZSBkaXJlY3RseSBhYm92ZSByaXNpbmcgbWFnbWEu[Qq]
[q] In the diagram below, a molecule of hydrogen (H2), still embedded in the crust in the ocean floor, is indicated, by what number? (Hint: look at water, H2O, at “1.”).
[textentry single_char=”true”]
[c]ID M=[Qq]
[f]IEV4Y2VsbGVudC4gJiM4MjIwOzMmIzgyMjE7IHJlcHJlc2VudHMgaHlkcm9nZW4gZ2FzLCBzdGlsbCB3aXRoaW4gdGhlIG9jZWFuIGZsb29yLg==[Qq]
[c]IEVudGVyIHdvcmQ=[Qq]
[f]IE5vLg==[Qq]
[c]ICo=[Qq]
[f]IE5vLiBIZXJlJiM4MjE3O3MgYSBoaW50LiBMb29rIGFnYWluIGF0IHRoZSB3YXRlciBtb2xlY3VsZSBhdCAmIzgyMjA7MS4mIzgyMjE7IFdoYXQmIzgyMTc7cyB0aGUgY29sb3Igb2YgdGhlIGh5ZHJvZ2VuIGF0b21zPw==[Qq]
[q] The lattice of iron, sulfur, and nickel shown below could have acted to [hangman] bonds between molecules at the vent, creating more complex monomers, and connecting the monomers to form [hangman].
[c]IGNhdGFseXpl[Qq]
[c]IHBvbHltZXJz[Qq]
[q multiple_choice=”true”] Which idea below is connected with the hypothetical mineral compartments that would have formed in alkaline hydrothermal vents?
[c]IGVhY2ggdmVudCBjb3VsZCBoYXZlIHJlcHJvZHVjZWQgaXRzZWxmLCBzcHJlYWRpbmcgdmVudHMgb3ZlciB0aGUgb2NlYW4gZmxvb3Iu[Qq]
[f]IE5vLiBWZW50cyBhcmUgc2l0dWF0ZWQgb25seSB3aGVyZSB0aGVyZSBpcyB1cHdlbGxpbmcgaGVhdCBhbmQgbWFnbWEuIFRoZXkgY2FuJiM4MjE3O3QgcmVwcm9kdWNlIHRoZW1zZWx2ZXMu[Qq]
[c]VGhlIHN0cnVjdHVyZSBvZiB0aGUgdmVudHMgd291bGQgbGVuZCBpdHNlbGYgdG8gY29uY2VudH JhdGluZyBtb25vbWVycywgZmFjaWxpdGF0aW5nIHRoZSBmb3JtYXRpb24gb2YgcG9seW1lcnMu[Qq]
[f]RXhhY3RseS4gVW5saWtlIHRoZSAmIzgyMjA7d2FybSBsaXR0bGUgcG9uZCYjODIyMTsgc2NlbmFyaW8sIGFsa2FsaW5lIGh5ZHJvdGhlcm1hbCB2ZW50cyBwcm92aWRlIGEgc3RydWN0dXJlIGZvciBjb25jZW50cmF0aW5nIG1vbm9tZXJzIGFuZCBmYWNpbGl0YXRpbmcgcG9seW1lciBmb3JtYXRpb24u[Qq]
[q labels = “top”]
| Alkaline hydrothermal Vents | Black smoker vents | |
| temperature | ___________ | __________ |
| pH | __________ | ___________ |
| vent structure | _______________ | ______________ |
| releases H2? | _______ | _______ |
| Age of vents | ______________ | ______________ |
| Location | ______________ | ___________________ |
| Magma powered? | ________________ | ______________ |
| Sun powered? | __________ | _________ |
[l] acidic
[fx] No, that’s not correct. Please try again.
[f*] Good!
[l] basic
[fx] No. Please try again.
[f*] Excellent!
[l]chimney-like
[fx] No, that’s not correct. Please try again.
[f*] Correct!
[l]complex, porous
[fx] No. Please try again.
[f*] Correct!
[l]Above ocean ridges
[fx] No, that’s not correct. Please try again.
[f*] Good!
[l]100s of years
[fx] No. Please try again.
[f*] Excellent!
[l]40°C to 90°C
[fx] No, that’s not correct. Please try again.
[f*] Excellent!
[l]400°C
[fx] No. Please try again.
[f*] Great!
[l]Off ridge
[fx] No. Please try again.
[f*] Correct!
[l]no
[fx] No, that’s not correct. Please try again.
[f*] Great!
[l]10,000s of years
[fx] No. Please try again.
[f*] Good!
[l]yes
[fx] No, that’s not correct. Please try again.
[f*] Great!
[/qwiz]
7. Links
In the next tutorial, we’ll look at the origins of heredity (the “RNA World”) the making of the first cells, and some issues related to metabolism. Then we’ll return to the vents.
- The RNA world, and Making Cells (the next tutorial in this module)
- Origin of Life, Main Menu
