1. Undersea hydrothermal vents and “metabolism first” chemical evolution
The “metabolism first” camp of origin-of-life-research promotes the idea that organized chains of chemical reactions — not replication — were the initial systems that led living systems to emerge. In other words, life started from a system that was conceptually similar to what’s shown: a linked series of natural flow reactors, where chemical reactions run in a continuously flowing stream.
Undersea hydrothermal vents, which were first discovered in the 1970s, are natural flow reactors,

Found at depths of 2500 meters below the surface of the ocean (and even deeper), undersea hydrothermal vents provide the energy and matter inputs that support dense ecosystems.
These systems are powered by molten magma rising toward the ocean floor. Seawater percolates into the ocean floor, becomes superheated, and then rises full of dissolved minerals. Deposition of these minerals forms natural chimneys that emit a continuous stream of black, smokey water (see the photo at left). As a result, these vents are called “Black Smokers.”
At the base of this community’s food web are bacteria that can perform chemosynthesis. Chemosynthesis parallels photosynthesis but uses hydrogen sulfide (H2S) instead of water (H2O).
The carbohydrate formed from these heat-powered reactions sustains communities of giant tube worms, shrimp, clams, mussels, and crabs, each of which has some type of relationship with the chemosynthetic bacteria. If you want to explore the biology of these Black Smoker Vents in more detail, click the following link for an amazing video with science journalist Ed Yong about the biology of vent tube worms.
Upon their discovery, origin-of-life researchers began speculating that ancient hydrothermal vents, with their constant flow of energy and their sheltered location — far away from the tides and meteor strikes that were occurring at the early Earth’s surface — could have been where life emerged. In fact, the chemistry of these vents is what inspired the hydrogen-sulfide world hypothesis about life’s origins that we briefly mentioned in the last tutorial.
However, a second type of undersea vent, discovered in the year 2000, has properties that would have been even more favorable for serving as a site for the origin of life. These are called alkaline hydrothermal vents, and here’s how they work.
2. Alkaline hydrothermal vents
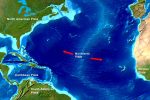
Alkaline Hydrothermal vents are also powered by geothermal energy, but less directly than the Black Smoker vents described above. The first of these vents to be discovered, called the Lost City, is located about 20 kilometers away from the mid-Atlantic ridge. The mid-Atlantic ridge is an undersea mountain range where new ocean crust is being created by upwelling magma.
The Lost City was discovered by a team from the University of Washington, led by Professor Deborah Kelley (the link goes to a page about Professor Kelley from the National Oceanic and Atmospheric Agency).

In the diagram on the left, you can see the overall geography of an alkaline hydrothermal vent. Number 1 shows rising magma, which is what causes seafloor spreading and creates structures like the mid-Atlantic Ridge and its rift valley (shown at number “3”). Number “2” is a black smoker vent. Off to the side, at number “4,” is an alkaline hydrothermal vent.

Like black smoker vents, minerals released at the Lost City vents create chimney-like structures. Spires composed of calcium carbonate grow to be several dozens of meters tall. Water leaving the vents is hot (40° C to 90° C), but much cooler than the 400°C fluid emerging from Black Smoker vents.
As their name indicates, the fluids emerging from these vents are alkaline (pH 9 to 11, as opposed to the slightly acidic fluid from Black Smoker Vents). These fluids are rich in highly reduced compounds, including molecular hydrogen (H2) and methane (CH4). The Lost City spires are also much longer lived than the Black Smoker chimneys. Radiometric dating sets the age of the Lost City system as at least 30,000 years old, making these vents hundreds of times older than Black Smoker Chimneys.
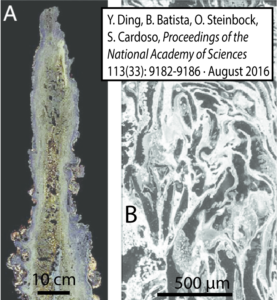
Another important difference between alkaline hydrothermal vents and Black Smoker vents is their internal structure. Alkaline hydrothermal vents are riddled with tiny interconnected pores, some of which are as small as a micrometer. As we’ll see in a moment, this scale is within the size range of bacterial cells, which range from 0.5 to 5 micrometers in diameter.
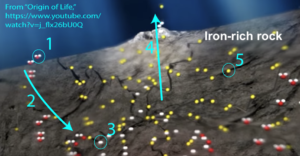
Because of all of these features, alkaline hydrothermal vents are increasingly viewed as an ideal ancient flow reactor where the abiotic synthesis of both monomers and polymers could have led to the emergence of living systems. Here’s how.
1. At these vents, the iron-rich sea floor chemically reacts with seawater (“1” in the diagram on the right). This reaction splits water molecules apart and results in the release of hydrogen gas (H2, shown at number “3”). Hydrogen gas is rare on Earth, yet it’s constantly released from alkaline vents.
The production of hydrogen gas is essential because it would have given emerging living systems one of the key outputs of photosynthesis — highly reduced compounds — without requiring highly structured membrane-bound machinery involved in photosynthesis. At alkaline hydrothermal vents, the hydrogen (along with its chemical energy and reducing power) is provided for free. And once you have reducing power, you’ve set the stage for the production of the reduced monomers that make up living systems.
2. At thousands of meters below the surface of the ocean, these vents would have provided a sheltered, stable environment, safe from the heavy bombardment the Earth was experiencing in Archaean times.
3. The flow of heat and hydrogen gas provided a consistent flow of gentle energy (which parallels the flow of sunlight that powers life today).
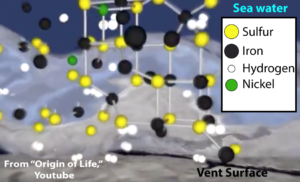
4. Unlike today’s oceans, the early oceans had high amounts of dissolved iron. The dissolved iron in seawater, along with other metals like nickel, would have combined with sulfur, creating iron-sulfur clusters. These clusters have catalytic properties, and often form the catalytic core of key enzymes that are still at work in living cells today.
5. These catalytic metals could have promoted the combination of the hydrogen gas emanating from the vents with carbon dioxide dissolved in the seawater, creating reduced carbon compounds like methane (CH4) or acetate (C2H3O2–). Further catalytic reactions could promote the formation of more complex molecules.

6. Finally, the porous structure of the alkaline vents would create microscopic chambers that could concentrate these monomers, promoting polymer formation. The formation of polymers would be further promoted by catalytic minerals lining the pores within the vents.
3. Origin of Life Video: Mike Russell and Bill Martin
Watch the video below, which pulls all of this together. You might want to watch it several times.
4. Flashcards: Origin of life at alkaline hydrothermal vents.
[qdeck bold_text=”false” style=”width: 650px !important; min-height: 450px !important;” qrecord_id=”sciencemusicvideosMeister1961-Origin of Life at Alkaline Hydrothermal Vents (v2.0)”]
[h] Origin of Life at Alkaline Hydrothermal Vents
[i]
[q] What’s the key idea of the “metabolism-first” camp?
[a] The “metabolism-first” camp promotes the idea that organized chains of chemical reactions were the initial systems that led to the emergence of living systems.
[q] Briefly describe “Black Smoker” vent ecosystems
[a] “Black smoker vents” are powered by rising magma. The magma superheats ocean water in the ocean floor, which rises full of dissolved minerals. Mineral deposition leads chimneys to form. The chimneys emit a rich, black smokey fluid (which is why these systems are called “Black Smokers.”
The food webs in these communities are based on chemosynthetic bacteria, which can use hydrogen sulfide as an electron and proton donor to create carbohydrates. The community supports worms, clams, crabs, shrimp, and other organisms.
[q]How are alkaline hydrothermal vents different from “Black smoker vents?”
[a]Alkaline hydrothermal vents are much cooler than Black smoker vents. They emit fluids that are alkaline, and rich in hydrogen. Their spires last much longer than “Black smoker” chimneys. Their internal structure is riddled with tiny pores.
[q]List four reasons why ancient alkaline hydrothermal vents are a promising site for a “metabolism first” origin of life.
[a]Ancient alkaline hydrothermal vents
- Naturally produce hydrogen gas, a rich energy source.
- Exist in an environment that would have been a stable refuge for life during the hellish Hadean era when life first emerged.
- Contain spires that are riddled with tiny pores that could have acted as reaction chambers, concentrating monomers into polymers.
- Would have produced metal catalysts that would promote the formation of monomers and polymers.
[x][restart]
[/qdeck]
5. Alkaline Hydrothermal Vents: Checking Understanding
[qwiz style=”width: 650px !important; min-height: 450px !important;” qrecord_id=”sciencemusicvideosMeister1961-Alkaline Hydrothermal Vents Quiz (v2.0)”]
[h]Alkaline Hydrothermal Vents: Checking Understanding
[i]
[q] Both “black smoker” vents and alkaline hydrothermal vents are powered by molten [hangman] rising toward the ocean floor.
[c]IG1hZ21h[Qq]
[f]IEdvb2Qh[Qq]
[q multiple_choice=”true”] Of the two hydrothermal vents discussed above, the type with hotter temperatures are
[c]IGFsa2FsaW5lIGh5ZHJvdGhlcm1hbCB2ZW50cw==[Qq]
[f]IE5vLiBUaGUgYWxrYWxpbmUgdmVudHMgYXJlIHNpZ25pZmljYW50bHkgY29vbGVyIHRoYW4gQmxhY2sgU21va2VyIHR5cGUgdmVudHM=[Qq]
[c]IEJsYWNrIHNtb2 tlciB2ZW50cw==[Qq]
[f]IEV4Y2VsbGVudC4gVGhlIGVmZmx1ZW50IGF0IGJsYWNrIHNtb2tlciB2ZW50cyBjYW4gYmUgdXAgdG8gNDAwIEM=[Qq]
[q] Black smoker-type vents support a food chain that is not based on photosynthesis, but on [hangman].
[c]IGNoZW1vc3ludGhlc2lz[Qq]
[f]IEdvb2Qh[Qq]
[q] At alkaline hydrothermal vents, a chemical reaction with rock (called serpentinization) causes the release of [hangman] gas into the seawater surrounding the vents.
[c]IGh5ZHJvZ2Vu[Qq]
[f]IENvcnJlY3Qh[Qq]
[q] In the diagram below, an alkaline hydrothermal vent is indicated by number
[textentry single_char=”true”]
[c]ID Q=[Qq]
[f]IENvcnJlY3QhwqA=TnVtYmVyIDQgc2hvd3MgYW4gYWxrYWxpbmUgdmVudC4=[Qq]
[c]IEVudGVyIGEgd29yZA==[Qq]
[f]IE5vLCB0aGF0JiM4MjE3O3Mgbm90IGNvcnJlY3Qu[Qq]
[c]ICo=[Qq]
[f]IE5vLiBOb3RlIHRoYXQgdGhlc2UgYWxrYWxpbmUgaHlkcm90aGVybWFsIHZlbnRzIGFyZSBhbHNvIGtub3duIGFzICYjODIyMDtvZmYgcmlkZ2UmIzgyMjE7IHZlbnRzLg==[Qq]
[q] In the diagram below, a black smoker-type vent is indicated by number
[textentry single_char=”true”]
[c]ID I=[Qq]
[f]IEdvb2Qh[Qq]
[c]IEVudGVyIHRoZSB3b3Jk[Qq]
[f]IE5vLCB0aGF0JiM4MjE3O3Mgbm90IGNvcnJlY3Qu[Qq]
[c]ICo=[Qq]
[f]IE5vLiBUaGUgYmxhY2sgc21va2VyIHZlbnRzIGFyZSBkaXJlY3RseSBhYm92ZSByaXNpbmcgbWFnbWEu[Qq]
[q] In the diagram below, a molecule of hydrogen (H2), still embedded in the crust on the ocean floor, is indicated by what number? (Hint: look at water, H2O, at “1.”).
[textentry single_char=”true”]
[c]ID M=[Qq]
[f]IEV4Y2VsbGVudC4gJiM4MjIwOzMmIzgyMjE7IHJlcHJlc2VudHMgaHlkcm9nZW4gZ2FzLCBzdGlsbCB3aXRoaW4gdGhlIG9jZWFuIGZsb29yLg==[Qq]
[c]IEVudGVyIHRoZSB3b3Jk[Qq]
[f]IE5vLg==[Qq]
[c]ICo=[Qq]
[f]IE5vLiBIZXJlJiM4MjE3O3MgYSBoaW50LiBMb29rIGFnYWluIGF0IHRoZSB3YXRlciBtb2xlY3VsZSBhdCAmIzgyMjA7MS4mIzgyMjE7IFdoYXQmIzgyMTc7cyB0aGUgY29sb3Igb2YgdGhlIGh5ZHJvZ2VuIGF0b21zPw==[Qq]
[q] The lattice of iron, sulfur, and nickel shown below could have acted to [hangman] bonds between molecules at the vent, creating monomers, and connecting the monomers to form [hangman].
[c]IGNhdGFseXpl[Qq]
[c]IHBvbHltZXJz[Qq]
[q multiple_choice=”true”] Which idea below is connected with the hypothetical mineral compartments that would have formed in alkaline hydrothermal vents?
[c]IGVhY2ggdmVudCBjb3VsZCBoYXZlIHJlcHJvZHVjZWQgaXRzZWxmLCBzcHJlYWRpbmcgdmVudHMgb3ZlciB0aGUgb2NlYW4gZmxvb3Iu[Qq]
[f]IE5vLiBWZW50cyBhcmUgc2l0dWF0ZWQgb25seSB3aGVyZSB0aGVyZSBpcyB1cHdlbGxpbmcgaGVhdCBhbmQgbWFnbWEuIFRoZXkgY2FuJiM4MjE3O3QgcmVwcm9kdWNlIHRoZW1zZWx2ZXMu[Qq]
[c]VGhlIHN0cnVjdHVyZSBvZiB0aGUgdmVudHMgd291bGQgbGVuZCBpdHNlbGYgdG8gY29uY2VudH JhdGluZyBtb25vbWVycywgZmFjaWxpdGF0aW5nIHRoZSBmb3JtYXRpb24gb2YgcG9seW1lcnMu[Qq]
[f]RXhhY3RseS4gQWxrYWxpbmUgaHlkcm90aGVybWFsIHZlbnRzIHByb3ZpZGUgYSBzdHJ1Y3R1cmUgZm9yIGNvbmNlbnRyYXRpbmcgbW9ub21lcnMgYW5kIGZhY2lsaXRhdGluZyBwb2x5bWVyIGZvcm1hdGlvbi4=[Qq]
[q labels = “top”]
| Alkaline hydrothermal Vents | Black smoker vents | |
| temperature | ______________ | __________ |
| pH | __________ | ___________ |
| vent structure | __________________ | ________________ |
| releases H2? | _______ | _______ |
| Age of vents | _________________ | _________________ |
| Location | ________________ | _____________________ |
| Magma powered? | ________________ | ______________ |
| Sun powered? | __________ | _________ |
[l] acidic
[fx] No, that’s not correct. Please try again.
[f*] Good!
[l] basic
[fx] No. Please try again.
[f*] Excellent!
[l]chimney-like
[fx] No, that’s not correct. Please try again.
[f*] Correct!
[l]complex, porous
[fx] No. Please try again.
[f*] Correct!
[l]Above ocean ridges
[fx] No, that’s not correct. Please try again.
[f*] Good!
[l]100s of years
[fx] No. Please try again.
[f*] Excellent!
[l]40°C to 90°C
[fx] No, that’s not correct. Please try again.
[f*] Excellent!
[l]400°C
[fx] No. Please try again.
[f*] Great!
[l]Off ridge
[fx] No. Please try again.
[f*] Correct!
[l]no
[fx] No, that’s not correct. Please try again.
[f*] Great!
[l]10,000s of years
[fx] No. Please try again.
[f*] Good!
[l]yes
[fx] No, that’s not correct. Please try again.
[f*] Great!
[/qwiz]
6. Origins of Life: Cumulative Flashcards
[qdeck style=”width: 650px !important; min-height: 450px !important;” random = “false” bold_text=”false” qrecord_id=”sciencemusicvideosMeister1961-Origin of Life: Cumulative Flashcards (v2.0)”]
[h]Origin of Life: Cumulative Flashcards
[i]
[q json=”true” yy=”4″ dataset_id=”AP_Bio_Flashcards_2022|14184585d8110″ question_number=”341″ unit=”7.Evolution_and_Natural_Selection” topic=”7.13.Origin_of_Life”] When did life first evolve on Earth? What’s the evidence?
[a] There’s widespread evidence for life on Earth as long ago as 3.4 billion years ago (bya), and life might have emerged even earlier than that. The consensus date for the emergence of life is about 3.8 bya.
The evidence for this claim consists of 1) fossilized bacterial mats (stromatolites), 2) microfossils of bacterial cells and 3) chemical traces of what’s called “biogenic carbon” (carbon of biological origin), all of which date back to 3.4 bya (or earlier).
[q json=”true” yy=”4″ unit=”7.Evolution_and_Natural_Selection” dataset_id=”AP_Bio_Flashcards_2022|140a02bd03910″ question_number=”342″ topic=”7.13.Origin_of_Life”] Describe the fossilized stromatolites and fossilized cells that serve as evidence for the earliest traces of life on Earth.
[a]
Stromatolites are layered bacterial mats, consisting of layers of cells interleaved with layers of sediments. These are formed in some areas on Earth today (such as Shark Bay in Australia). The oldest fossilized stromatolites have been dated (through radiometric dating) as 3.4 billion years old.
Inside these fossilized stromatolites are microfossils. These are fossils that are cellular in size, and can only be visualized with an electron microscope. When ancient fossil stromatolites are sectioned and viewed under an electron microscope, structures that match cells in size and form can be seen.
[q json=”true” yy=”4″ unit=”7.Evolution_and_Natural_Selection” dataset_id=”AP_Bio_Flashcards_2022|13fc7a37e6510″ question_number=”343″ topic=”7.13.Origin_of_Life”]Isotopic signatures can serve as evidence for the earliest traces of life on Earth. Explain.
Illustrative example. Evidence for the origin of life
[a] When living things take in carbon, they preferentially take in carbon-12 over a heavier (and much rarer) isotope, carbon-13. In carbon that’s biological in origin, the ratio of carbon 12 to carbon 13 is much higher than in carbon samples of non-biological origin. Samples of carbon from 3.4 billion years ago (and even up to 3.8 billion years ago) show that this carbon was captured by living things. Note that using isotopic signatures alone, claims have been made for an origin of life that occurred over 4 billion years ago, but these are not widely accepted.
[q json=”true” yy=”4″ dataset_id=”AP_Bio_Flashcards_2022|13ef16f387510″ question_number=”344″ unit=”7.Evolution_and_Natural_Selection” topic=”7.13.Origin_of_Life”] It’s been said that life emerged on Earth about as early as it possibly could have. Explain.
[a]Our solar system, including our Earth, formed about 4.6 bya from a stellar nebula. As the planets formed, there was a period of massive collisions when the early Earth was under continual bombardment by comets, asteroids, and even planet-sized objects. These bombardments would have made life impossible. Yet signs of life are unambiguous at 3.4 bya, and life might have evolved as early as 3.8 bya, which is close to when the period of heavy bombardment was coming to a close. Hence, life emerged on our world just about as early as it could have.
[q json=”true” yy=”4″ unit=”7.Evolution_and_Natural_Selection” topic=”7.13.Origin_of_Life” dataset_id=”AP_Bio_Flashcards_2022|1781950e37dcd5″ question_number=”345″] Describe some the conditions on the early Earth during the period when life first emerged.
[a]Life on Earth emerged when conditions were quite different from conditions today. First of all, Earth had very little land: just a few islands above a world ocean. The young moon was much closer, causing massive tides in these oceans. The Earth spun more quickly on its axis: a day might have been as short as eight hours. The atmosphere was different, consisting of carbon dioxide and nitrogen, and no molecular oxygen (O2).
[q json=”true” yy=”4″ dataset_id=”AP_Bio_Flashcards_2022|13e1d8efe6910″ question_number=”346″ unit=”7.Evolution_and_Natural_Selection” topic=”7.13.Origin_of_Life”] In any origin of life scenario, what’s the first step? Explain.
[a] In any origin of life scenario, the first step is the formation of monomers: the amino acids, nucleotides, monosaccharides, and fatty acids that serve as the building blocks of life. Only if these molecules are present can more complex molecules (like proteins and nucleic acids) come into being.
Monomer formation is difficult to explain because these monomers don’t spontaneously form. Rather, these energy-rich molecules are generated biologically by plants, cyanobacteria, and algae during photosynthesis. So the challenge in explaining the origin of life is to explain how to generate monomers abiotically (in the absence of life).
[q json=”true” yy=”4″ unit=”7.Evolution_and_Natural_Selection” dataset_id=”AP_Bio_Flashcards_2022|13d888c056910″ question_number=”347″ topic=”7.13.Origin_of_Life”] The Miller-Urey experiment was an attempt to validate Oparin and Haldane’s idea, proposed in the 1920s, of monomers emerging in a primordial soup. What was the primordial soup?
[a] The primordial-soup model posits that in the Earth’s early oxygen-free atmosphere, available energy could cause molecules like methane and ammonia to combine to form monomers (monosaccharides, amino acids, etc.), which would accumulate in the early oceans, forming a primordial soup. This, in turn, would set the stage for more complex molecules to form, leading to the emergence of life.
[q json=”true” yy=”4″ unit=”7.Evolution_and_Natural_Selection” topic=”7.13.Origin_of_Life” dataset_id=”AP_Bio_Flashcards_2022|d112b223e40ea” question_number=”348″] Describe the Miller-Urey experiment and its results.
[a]The Miller-Urey experiment validated the idea of the abiotic formation of monomers. The experiment combined methane, ammonia, and hydrogen (thought at that time to be a plausible mix for the Early earth’s atmosphere) in a reaction chamber (5). The chamber was connected to a flask containing heated water (2) which represented the early Earth’s oceans. Sparks (5) simulated lightning. After several days, the circulating mixture in the apparatus was sampled, and analysis showed the presence of several amino acids (the monomers of proteins).
[q json=”true” yy=”4″ unit=”7.Evolution_and_Natural_Selection” dataset_id=”AP_Bio_Flashcards_2022|13c404178d510″ question_number=”349″ topic=”7.13.Origin_of_Life”] Why is the Miller-Urey experiment widely regarded as a keystone in origin of life research, and why has it been widely criticized?
[a] The overall achievement of the Miller-Urey experiment was validation of the idea that monomers can be formed abiotically. In subsequent years, this has been confirmed in a variety of experimental settings, with many different starting compounds and energy sources. This idea has also been validated by the discovery of monomers on meteorites (meaning that certain monomers can form out in space). However, the experiment has been criticized for using an atmosphere with highly reduced gases (methane, ammonia, and hydrogen), which are now thought to have been unlikely on the early Earth, and which made it much easier to produce compounds such as amino acids.
[q json=”true” yy=”4″ unit=”7.Evolution_and_Natural_Selection” topic=”7.13.Origin_of_Life” dataset_id=”AP_Bio_Flashcards_2022|d0f5978f420ea” question_number=”350″] The Miller-Urey experiment was an attempt to validate the “primordial soup” model of monomer formation. But while life requires water, the presence of water is also a problem in any origin of life scenario. Explain.
[a]The problem is that the polymerization of monomers to create polymers such as RNA and protein would be difficult in a watery environment. That’s because monomers combine to form polymers through dehydration synthesis, a reaction that only occurs in a watery solution with the help of enzymes. Without enzymes (which are themselves polymers), it’s hard to imagine how polymer formation could have happened. In other words, life’s first nursery was unlikely to have been any type of soupy, open body of water. Rather, a system like a hydrothermal vent is a much more likely venue for the origin of life.
[q json=”true” yy=”4″ dataset_id=”AP_Bio_Flashcards_2022|13bafe6979d10″ question_number=”351″ unit=”7.Evolution_and_Natural_Selection” topic=”7.13.Origin_of_Life”] How was the early earth’s atmosphere different from the atmosphere today?
[a] The Earth’s earliest atmosphere (like that of Mars and Venus) lacked molecular oxygen (O2). That’s because O2 only exists on Earth because of photosynthesizing organisms that oxidize water and release O2 as a waste product. Thus, free O2, which makes up about 21% of the atmosphere today, is a planetary-scale biogenic signature: a feature that arises only in the presence of life. If in the future, a space probe finds itself orbiting a planet with an O2-rich atmosphere, we’ll be fairly certain that this is a planet where life (at least microbial life) is flourishing (even before our probe lands and takes samples).
[q json=”true” yy=”4″ unit=”7.Evolution_and_Natural_Selection” dataset_id=”AP_Bio_Flashcards_2022|13abb6db71910″ question_number=”352″ topic=”7.13.Origin_of_Life”] The early Earth’s atmosphere was quite different from the atmosphere today. Why is that important for the origin of life?
[a] Unlike today’s atmosphere, which consists of about 21% oxygen, the early Earth’s atmosphere has no free oxygen (O2). The reason why this is important to the origin of life is that for life to arise, its molecular components, biological monomers, need to arise. These monomers are reduced molecules, and in the presence of oxygen, their spontaneous tendency is to become oxidized. A world with abundant oxygen is not a world where new life could arise, and the presence of a reducing (or at least non-oxidizing) atmosphere and ocean was probably a precondition for the emergence of the monomers (amino acids, nucleotides, fatty acids, and monosaccharides) that were combined to create the polymers in the first living cells.
[q json=”true” yy=”4″ dataset_id=”AP_Bio_Flashcards_2022|139bff8b2e910″ question_number=”353″ unit=”7.Evolution_and_Natural_Selection” topic=”7.13.Origin_of_Life”] What is the RNA world hypothesis?
[a] The RNA world hypothesis is the idea that the first genetic molecule was RNA, not DNA. That’s because DNA is too protein-dependent for its replication and transcription to have arisen on its own. Without proteins (like DNA polymerases), you can’t replicate DNA. But polymerases are proteins and are spelled out by DNA. How could we have one arise without the other? RNA is thought to be a better candidate as the first genetic molecule because RNA can be both information (as in messenger RNA) and act as a catalyst (as in ribosomal RNA). According to the theory, if a self-replicating RNA could arise, then that would set the stage for the emergence of evolution and life.
[q json=”true” yy=”4″ unit=”7.Evolution_and_Natural_Selection” dataset_id=”AP_Bio_Flashcards_2022|13918555ac910″ question_number=”354″ topic=”7.13.Origin_of_Life”] What are two attributes of RNA that make it a better candidate than DNA for playing the role of the first genetic molecule?
[a] RNA is thought to be a better candidate for the first genetic molecule than DNA because RNA can serve as both genetic information (as it does in RNA viruses and mRNA) and as a catalyst for chemical reactions (as it does in spliceosomes and as the catalytic part of ribosomes). DNA, by contrast, is purely informational.
[q json=”true” yy=”4″ unit=”7.Evolution_and_Natural_Selection” dataset_id=”AP_Bio_Flashcards_2022|13823dc7a4510″ question_number=”355″ topic=”7.13.Origin_of_Life”] Using the RNA-world hypothesis, sketch out the steps to the emergence of DNA-based and cell-based life.
[a] The RNA-world hypothesis posits that as life was arising, a population of self-replicating RNAs emerged (1 and 2). During self-replication, some errors would arise, leading to diverse RNAs that could be subject to natural selection. Those RNA replicators that could replicate more quickly and efficiently would come to dominate their populations. At some point, self-replicating RNA polymers became paired with amino acids and polypeptides, leading to the emergence of RNA-protein systems (3). Next, these self-replicating RNA-protein systems would have had DNA replace RNA as the genetic material (4). Finally, encapsulation by lipid membranes would lead to the first cells (5).
[x]
[restart]
[/qdeck]
7. Origins of Life: Cumulative Quiz
Let’s consolidate what we’ve learned with the following quiz. It includes information from the entire module.
[qwiz style=”width: 650px !important; min-height: 450px !important;” random = “false” qrecord_id=”sciencemusicvideosMeister1961-Origin of Life: Cumulative Quiz (v2.0)”]
[h]Origin of Life: Cumulative Quiz
[i]
[q labels = “top”]Describe the traits of LUCA.
- _________ celled _____________
- Genetic information was encoded in ______.
- Genes were transcribed into ______, which was translated into ___________ composed of 20 _______________.
- Used the universal genetic code, with 64 __________.
- Translation was catalyzed by____________.
- Metabolism created _________ to power cellular work.
- The entire cell was surrounded by a lipid _____________.
- Created ATP by ________________.
[l]amino acids
[f*] Correct!
[fx] No. Please try again.
[l]ATP
[f*] Excellent!
[fx] No, that’s not correct. Please try again.
[l]chemiosmosis
[f*] Great!
[fx] No, that’s not correct. Please try again.
[l]codons
[f*] Excellent!
[fx] No. Please try again.
[l]DNA
[f*] Excellent!
[fx] No. Please try again.
[l]membrane
[f*] Excellent!
[fx] No, that’s not correct. Please try again.
[l]prokaryote
[f*] Correct!
[fx] No. Please try again.
[l]proteins
[f*] Excellent!
[fx] No, that’s not correct. Please try again.
[l]ribosomes
[f*] Good!
[fx] No, that’s not correct. Please try again.
[l]RNA
[f*] Great!
[fx] No. Please try again.
[l]Single
[f*] Good!
[fx] No. Please try again.
[q multiple_choice=”true”] Which of the following is a widely accepted date for the origin of life?
[c]IDQuNSBieWE=[Qq]
[f]IE5vLiBUaGF0JiM4MjE3O3Mgd2F5IHRvbyBlYXJseS4gVGhhdCYjODIxNztzIHdoZW4gdGhlIEVhcnRoIHdhcyBmaXJzdCBmb3JtaW5nLg==[Qq]
[c]IDQuMyBieWE=[Qq]
[f]IE5vLiBUaGF0JiM4MjE3O3MgZHVyaW5nIHRoZSBwZXJpb2Qgb2YgaGVhdnkgYm9tYmFyZG1lbnQuIEFueSBlbWVyZ2luZyBsaWZlIHByb2JhYmx5IHdvdWxkIGhhdmUgYmVlbiBkZXN0cm95ZWQgYnkgaW5jb21pbmcgbWV0ZW9ycyBhbmQgY29tZXRzLg==[Qq]
[c]IDMuOC BieWE=[Qq]
[f]IEV4Y2VsbGVudCEgWW91JiM4MjE3O3ZlIGNob3NlbiBhIGRhdGUgdGhhdCYjODIxNztzIGJldHdlZW4gdGhlIGVuZCBvZiB0aGUgcGVyaW9kIG9mIGhlYXZ5IGJvbWJhcmRtZW50LCBhbmQgYmVmb3JlIHRoZSBkYXRlIGZvciB0aGUgZWFybGllc3QgZm9zc2lsaXplZCBsaWZlLiBMaWZlIGhhZCB0byBlbWVyZ2Ugc29tZXdoZXJlIGluIGJldHdlZW4u[Qq]
[c]IDMuNSBieWE=[Qq]
[f]IE5vLiBBcyBzaG93biBieSBmb3NzaWxpemVkIHN0cm9tYXRvbGl0ZXMgYW5kIGFzc29jaWF0ZWQgbWljcm9mb3NzaWxzIGZyb20gdGhhdCBwZXJpb2QsIGxpZmUgd2FzIGFscmVhZHkgd2VsbCBlc3RhYmxpc2hlZCBieSAzLjUgYnlhLiBDaG9vc2UgYW4gZWFybGllciBwZXJpb2Qu[Qq]
[q] Dating of ancient structures like fossilized stromatolites is done through a method that involves measuring the decay of radioactive [hangman] of certain elements, a method known as [hangman] dating.
[c]IGlzb3RvcGVz[Qq]
[c]IHJhZGlvbWV0cmlj[Qq]
[q] Stanley Miller’s experiments were significant because he successfully synthesized [hangman] acids in a simulation of the Earth’s early atmosphere.
[c]IGFtaW5v[Qq]
[q] During the Hadean/Archaean periods when life was first emerging, the Earth was very different from today’s Earth. The atmosphere lacked the gas [hangman]. The [hangman] was much closer, causing enormous tides. The Earth was in a period of heavy bombardment and suffered frequent impacts from [hangman] and comets.
[c]b3h5Z2Vu[Qq]
[c]IG1vb24=[Qq]
[c]IG1ldGVvcnM=[Qq]
[q]Alkaline [hangman] vents, which release[hangman]gas into the environment, might have provided an environment conducive to the evolution of life.
[c]IGh5ZHJvdGhlcm1hbA==
[c]IGh5ZHJvZ2Vu[Qq]
[q]The type of vent shown below is called a [hangman] [hangman] vent. Some origin-of-life researchers think that this type of vent is an unlikely candidate for life’s emergence because the water that’s emerging from them is too [hangman] to allow for the kind of chemistry that would have created the first monomers and polymers.
[c]YmxhY2s=[Qq]
[c]c21va2Vy[Qq]
[c]IGhvdA==[Qq]
[q]At alkaline hydrothermal vents, geological processes causes the release of [hangman] gas, which is otherwise relatively rare on Earth. When this gas is combined with carbon dioxide, it could have formed simple hydrocarbons. Because of inorganic [hangman] that build up in these vents, these hydrocarbons could have been assembled into the first [hangman], which would later combine into [hangman].
[c]IGh5ZHJvZ2Vu[Qq]
[c]Y2F0YWx5c3Rz[Qq]
[c]bW9ub21lcnM=[Qq]
[c]cG9seW1lcnM=[Qq]
[q]The first membrane-bound cells might have formed as an accumulation of [hangman] acids led to the formation of micelles. These micelles would aggregate together, forming fluid-filled spheres, the outer “skin” of which was a lipid [hangman].
[c]ZmF0dHk=[Qq]
[c]YmlsYXllcg==[Qq]
[q] If “c” in the diagram below represents RNA, then ribonucleotides would be represented by
[textentry single_char=”true”]
[c]Qg ==[Qq]
[f]IE5pY2UuIFRoZSBsZXR0ZXIgJiM4MjIwO2ImIzgyMjE7IHJlcHJlc2VudHMgdGhlIG1vbm9tZXJzIHRoYXQgbWFrZSB1cCBSTkEsIHdoaWNoIGFyZSByaWJvbnVjbGVvdGlkZXMu[Qq]
[c]IHdvcmQ=[Qq]
[f]wqA=Tm8u[Qq]
[c]ICo=[Qq]
[f]IE5vLiBMb29rIGZvciBtb25vbWVycyB0aGF0LCBpZiBzdHJ1bmcgdG9nZXRoZXIsIGNvdWxkIGNyZWF0ZSBhbiBSTkEgcG9seW1lciAoYXQgJiM4MjIwO0MmIzgyMjE7KS4=[Qq]
[q] In the diagram below, the only letter that could represent a ribozyme would be
[textentry single_char=”true”]
[c]RA ==[Qq]
[f]IE5pY2UuIExldHRlciAmIzgyMjA7ZCYjODIyMTsgc2hvd3MgUk5BIHRoYXQmIzgyMTc7cyBmb2xkZWQgaW50byBhIG1vcmUgY29tcGxleCAzLUQgc2hhcGUsIGFuZCB0aGF0JiM4MjE3O3MgaG93IGEgcmlib3p5bWUgd291bGQgZm9ybSAodGhvdWdoIGluIHRoaXMgcmVwcmVzZW50YXRpb24sIHRoZSBzaGFwZSBpcyB2ZXJ5IHNpbXBsZSku[Qq]
[c]IHdvcmQ=[Qq]
[f]wqA=Tm8u[Qq]
[c]ICo=[Qq]
[f]IE5vLiBMb29rIGZvciBhbiBSTkEgcG9seW1lciB0aGF0JiM4MjE3O3MgZm9sZGluZyBpbnRvIGEgbW9yZSBjb21wbGV4IHNoYXBlLg==[Qq]
[q multiple_choice=”true”] In terms of bringing about the RNA world, which of the following steps is the one that has the most experimental evidence?
[c]IEZyb20gQSB0byBC[Qq]
[f]IE5vLiBMZXR0ZXIgJiM4MjIwO2EmIzgyMjE7IHRvIGxldHRlciAmIzgyMjA7YiYjODIyMTsgc2hvd3MgKHZlcnkgc2NoZW1hdGljYWxseSkgdGhlIGNvbXBvbmVudHMgb2YgUk5BIChyaWJvc2UsIG5pdHJvZ2Vub3VzIGJhc2VzLCBhbmQgcGhvc3BoYXRlcykgY29taW5nIHRvZ2V0aGVyIHRvIGZvcm0gYSByaWJvbnVjbGVvdGlkZS4gVGhhdCYjODIxNztzIGJlZW4gc29tZXdoYXQgZGlmZmljdWx0IHRvIGJyaW5nIGFib3V0IGluIHRoZSBsYWJvcmF0b3J5Lg==[Qq]
[c]IEZyb20gQiB0byBD[Qq]
[f]IE5vLiBGb3JtaW5nIHRoZSAmIzgyMjA7YmFja2JvbmUmIzgyMjE7IG9mIFJOQSBpcyBub3Qgc29tZXRoaW5nIHRoYXQmIzgyMTc7cyBiZWVuIGV4cGVyaW1lbnRhbGx5IHJlcHJvZHVjZWQgaW4gYSBsYWJvcmF0b3J5IHNldHRpbmcu[Qq]
[c]IEZyb20g QyB0byBE[Qq]
[f]IEV4Y2VsbGVudCEgT25jZSBSTkEgbW9sZWN1bGVzIGhhdmUgZm9ybWVkLCBpdCYjODIxNztzIG5vdCBoYXJkIHRvIGdldCB0aGVtIHRvIGZvbGQgdXAgaW50byBhIHNwZWNpZmljIDMtRCBzaGFwZS4=[Qq]
[q] In the diagram below, lipids would play a key role in which step?
[textentry single_char=”true”]
[c]Zg ==[Qq]
[f]IE5pY2UuIExldHRlciAmIzgyMjA7ZiYjODIyMTsgc2hvd3MgUk5BIGVuY2Fwc3VsYXRlZCB3aXRoaW4gd2hhdCBsb29rcyBsaWtlIGEgY2VsbC4gVGhlIG1lbWJyYW5lIG9mIHRoYXQgY2VsbCB3b3VsZCBiZSBzb21lIGtpbmQgb2YgbGlwaWQu[Qq]
[c]IHdvcmQ=[Qq]
[f]wqA=Tm8u[Qq]
[c]ICo=[Qq]
[f]IE5vLiBSZW1lbWJlciB0aGF0IGEgY2VsbCBtZW1icmFuZSBpcyBjb21wb3NlZCBvZiBhIGxpcGlkIGJpbGF5ZXIuIFdoZXJlIGRvIHlvdSBzZWUgYSBjZWxsIG1lbWJyYW5lPw==[Qq]
[q] Fossilized, layered structures like the ones below are remnants of structure created by bacteria. These structures are called [hangman].
[c]IHN0cm9tYXRvbGl0ZXM=[Qq]
[f]IEdyZWF0IQ==[Qq]
[q] According to NASA, life is a self-[hangman] chemical system capable of Darwinian evolution.
[c]IHN1c3RhaW5pbmc=[Qq]
[f]IEdyZWF0IQ==[Qq]
[q]Isotope M has a half-life of 5000 years, during which it decays into isotope N. You’re studying a sample of material that has a relative proportion of 25% M and 75% N. How old is this material?
[c]IDUwMDAgeWVhcnMgb2xkLg==[Qq]
[f]IE5vLiA1MDAwIHllYXJzIGlzIG9uZSBoYWxmLWxpZmUsIGR1cmluZyB3aGljaCB0aW1lIHlvdSYjODIxNztkIGV4cGVjdCBoYWxmIG9mIE0gdG8gZGVjYXkgaW50byBOLCBmb3IgYSA1MC81MCByYXRpby4=[Qq]
[c]IDc1MCB5ZWFycy4=[Qq]
[f]IE5vLiBIZXJlJiM4MjE3O3MgaG93IHRvIHRoaW5rIGFib3V0IHRoaXMuIDUwMDAgeWVhcnMgaXMgb25lIGhhbGYtbGlmZSwgZHVyaW5nIHdoaWNoIHRpbWUgeW91JiM4MjE3O2QgZXhwZWN0IGhhbGYgb2YgTSB0byBkZWNheSBpbnRvIE4sIGZvciBhIDUwLzUwIHJhdGlvLiBCYXNlZCBvbiB0aGUgcXVlc3Rpb24sIGZpbmQgdGhlIG51bWJlciBvZiBoYWxmLWxpdmVzLCBhbmQgbXVsdGlwbHkgdGhhdCBieSB0aGUgZHVyYXRpb24gb2YgYSBoYWxmLWxpZmUu[Qq]
[c]IDEwLDAwMCB5 ZWFycyBvbGQ=[Qq]
[f]IEV4Y2VsbGVudC4gSWYgb25seSAyNSUgb2YgdGhlIG9yaWdpbmFsIG1hdGVyaWFsIGlzIGxlZnQsIHRoZW4gdHdvIGhhbGYtbGl2ZXMgaGF2ZSBwYXNzZWQsIGZvciBhIHRvdGFsIG9mIDEwLDAwMCB5ZWFycy4=[Qq]
[q]The proponents of the RNA world would fall into the [hangman]-first camp. The other camp, supported by research on the chemistry of undersea alkaline hydrothermal vents, is the [hangman]-first camp.
[c]Z2VuZXM=[Qq]
[c]bWV0YWJvbGlzbQ==[Qq]
[x]
[restart]
[/qwiz]
8. The Origins of Life’s Chemiosmotic Metabolism
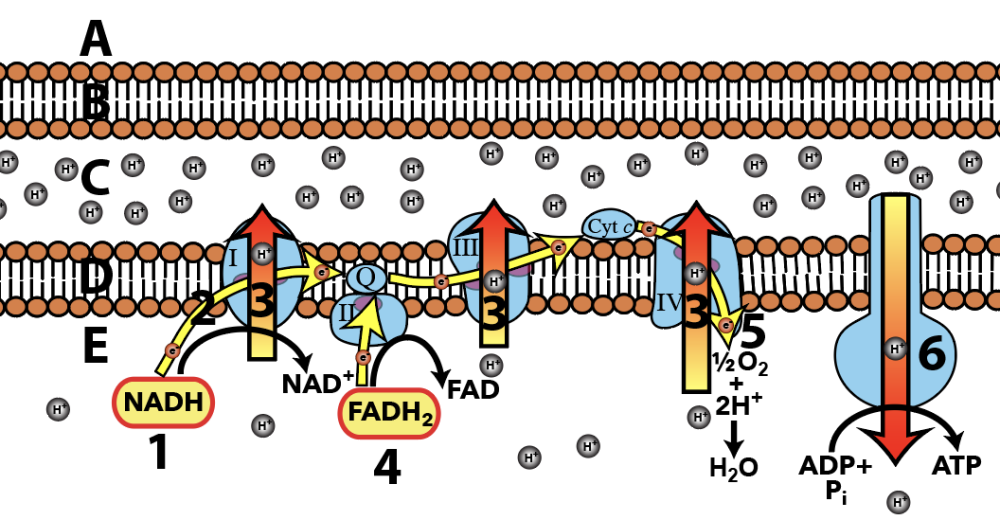
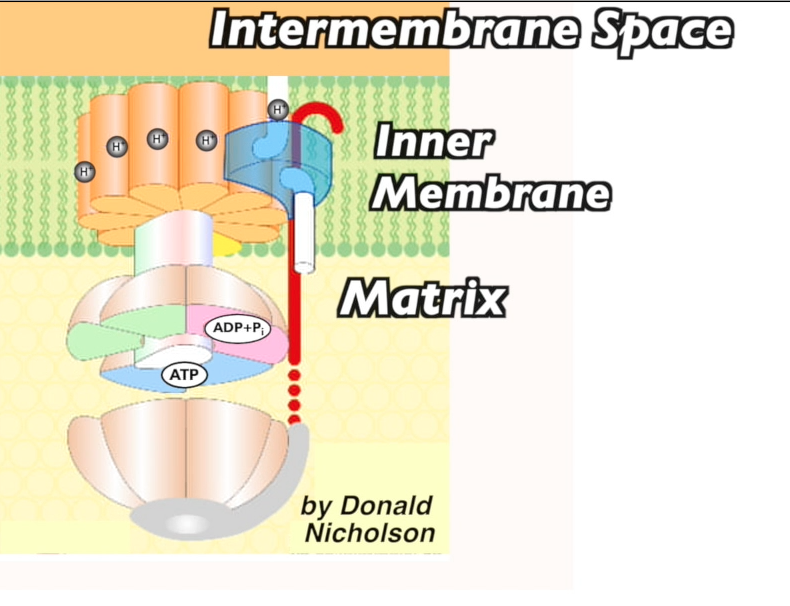
In all living organisms today, the energy source for creating ATP is based on chemiosmosis: protons diffusing through an ATP synthase channel embedded in a membrane. This feature of life is as universal as DNA and ribosomes and was almost certainly present in LUCA.
Here’s what chemiosmotic proton flow looks like in mitochondria, during cellular respiration.
And here’s what it looks like in chloroplasts, during photosynthesis.
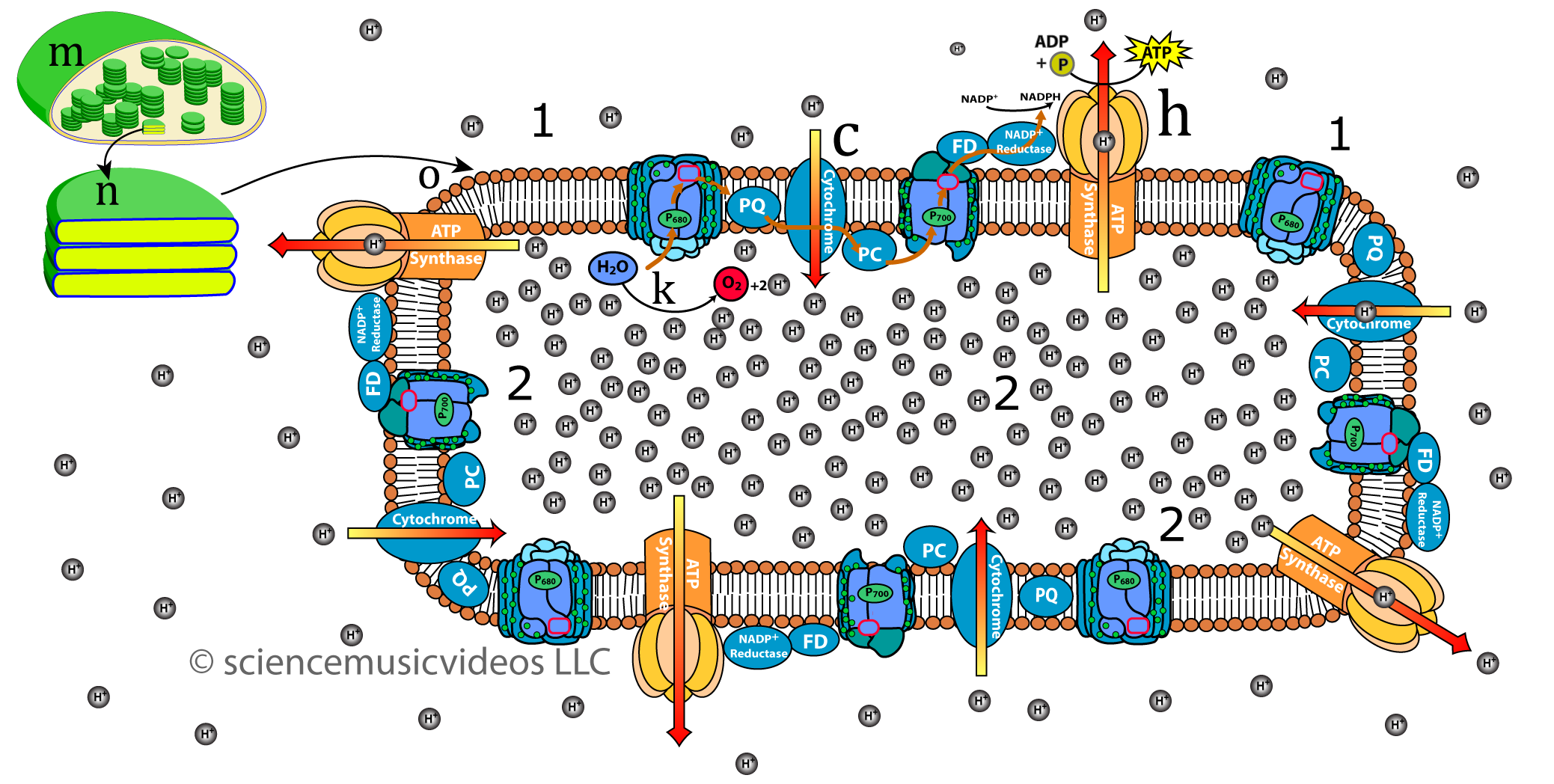
In both chloroplasts and mitochondria, proton flow through ATP synthase is based upon electron flow, which powers proton pumps.
- Chloroplast and cyanobacteria convert light energy into a flow of electrons. The energy from that flow is used to pump protons into the thylakoid spaces in thylakoid sacs. Then the protons diffuse out through ATP synthase, generating ATP.
- In mitochondria (and many bacteria and archaea), a flow of electrons from food molecules is used to pump protons into the mitochondrial intermembrane space. Then, just in photosynthesis, the protons flow out through ATP synthase, generating ATP.
ATP-creation by chemiosmosis is one of the deep homologies that unify all living things. So, assuming that life emerged not at the Earth’s sunlight surface, but deep in the ocean depths, how could ATP-creating proton gradients have evolved?
Let’s review what’s happening in alkaline hydrothermal vents about 3.8 bya. Just like today, alkaline fluids would be rising from the sea floor and percolating into the tiny chambers that form within these vents. Unlike today’s seas, which are slightly basic (pH 8.1), the surrounding ancient seawater would have been much more acidic. That’s because ocean water had much more dissolved carbon dioxide, which dissolves in water to become carbonic acid. In the subsequent billions of years, much of that CO2 has been removed by photosynthesis, raising the ocean’s pH to its current level.
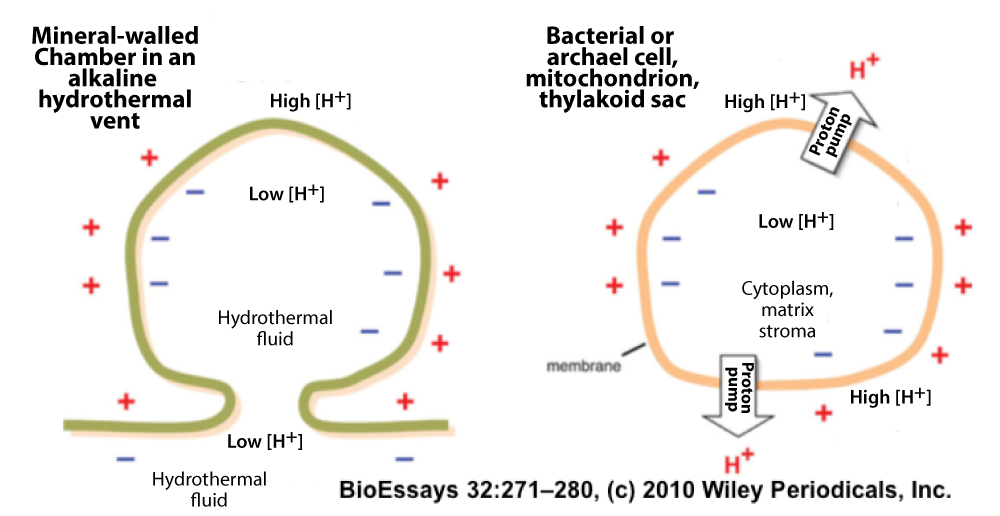
The image on the left shows one of these tiny chambers on the left side, with a cell shown on the right for comparison. Alkaline fluids, created geologically in the rocks below the vents (notice the “Low [H]” label below the green inorganic chamber on the left) would have flowed into these chambers. This would have created a natural proton gradient between the acidic seawater outside the vents and the alkaline fluid inside the vents. That proton gradient is parallel to the one that cells today have to create by proton pumping that’s powered by food or sunlight. But in the vents, the hydrogen ion gradient was there for free. If there were tiny pores in these chambers, protons could flow “downhill” from outside these rocky chambers to the inside. This flow, if captured, could power the creation of phosphorylated compounds, forerunners of ATP. These compounds, in turn, could provide the chemical energy to power the creation of RNA polymers, leading to the RNA world…
Here’s a quote from biochemist Nick Lane, author of Life Ascending, one of my favorite biology books.
The last common ancestor of all life was not a free-living cell at all, but a porous rock riddled with bubbly iron-sulfur membranes that catalyzed primordial biochemical reactions. Powered by hydrogen and proton gradients, this natural flow reactor filled up with organic chemicals, giving rise to proto-life that eventually broke out as the first living cells… (Nick Lane, The Cradle of Life: New Scientist, 17, October 2009).
There are some big gaps in this account. One gap is the emergence of ATP synthase, a complex molecular machine with a spinning rotor and multiple interdependent parts. Origin of life researchers will have to continue their work to create a credible account of how this nano-machine could have arisen in a step-by-step manner.
9. Nick Lane’s Ten Steps to the emergence of Life on Earth
Nick Lane is a British biochemist whose work has been referenced several times in this module. He has done an amazing job popularizing the most recent advances related to the origins of life. He’s been enormously influential in shaping my understanding of living systems, and that, in turn, had deeply influenced my teaching.
Here are his Ten Steps to the Origin of Life (taken verbatim from an article he wrote in NewScientist | 17 October 2009).
If life did evolve in alkaline hydrothermal vents, it might have happened something like this:
1. Water percolated down into newly formed rock under the sea floor, where it reacted with minerals such as olivine, producing a hot alkaline fluid rich in hydrogen, sulfides, and other chemicals. This hot fluid welled up at hydrothermal vents.
2. The early ocean was acidic and rich in dissolved iron. When upwelling hydrothermal fluids reacted with this seawater, they produced carbonate rocks riddled with tiny pores and a “foam” of iron-sulfur bubbles.
3. Inside the bubbles, hydrogen reacted with carbon dioxide to form simple organic molecules such as methane, formate, and acetate. Some of these reactions were catalyzed by the iron-sulfur minerals, which are still found at the heart of many proteins today.
4. The electrochemical gradient between the alkaline vent fluid and the acidic seawater leads to the spontaneous formation of acetyl phosphate and pyrophosphate, which act just like ATP, the chemical that powers living cells. These molecules drove the formation of amino acids and nucleotides, the building blocks of proteins and of RNA and DNA respectively.
5. Thermal currents within the vent pores concentrated large molecules like nucleotides, driving the formation of RNA and DNA – and providing an ideal setting for an RNA world and its evolution into the world of DNA and proteins. Evolution got underway, with sets of molecules capable of producing more of themselves starting to dominate.
6. Fatty molecules coated the iron-sulfur froth and spontaneously formed cell-like bubbles. Some of these bubbles would have enclosed self-replicating sets of molecules – the first organic cells. The earliest protocells may have been elusive entities, though, often dissolving and reforming as they circulated in the vents.
7. The evolution of an enzyme called pyrophosphatase, which catalyzes the production of pyrophosphate, allows protocells to extract more energy from the gradient between the alkaline vent fluid and the acidic ocean. This enzyme is still found in many bacteria and archaea.
8. Some protocells start using ATP as well as acetyl phosphate and pyrophosphate. The production of ATP using energy from the electrochemical gradient is perfected with the evolution of the enzyme ATP synthase, found within all life today.
9. Protocells further from the main vent axis, where the natural electrochemical gradient is weaker, started to generate their own gradient to drive ATP production.
10. Once protocells could generate their own electrochemical gradient, they were no longer tied to the vents. Cells left the vents on two separate occasions, with one exodus giving rise to bacteria and the other to archaea.
10. Further Reading
If you want to take a deeper dive into this material, you can read articles by Russell, Martin, and some of their collaborators (including Deborah Kelley, who discovered alkaline hydrothermal vents in 2000), at this link. A much more accessible article written by Nick Lane can be found here. Another review of the pros and cons of alkaline hydrothermal vents can be found in this article in Chemistry World.
If you have the time and the inclination, please follow some of the suggested links below, each of which is linked to specific questions related to the origins of life. All links open new tabs (and note that some of this is very dense reading).
- How did the genetic code arise? See articles by Keeling, or Koonin and Novoshilov.
- How did the system of protein translation from mRNA evolve? See articles by Noller, or Petrov et.al.
- How and when did DNA take over from RNA? See Forterre et. al
11. What’s Next?
This tutorial ends Topic 7.12, The Origin of Life. It also ends Unit 7, Evolution. To review and prepare for your unit assessment, proceed to these Unit 7 Cumulative Objectives, Flashcards, and Quizzes.
